Show that, in principle, Na 2 CO 3 (aq) can be converted almost completely to NaOH(aq) by
Question:
Show that, in principle, Na2CO3(aq) can be converted almost completely to NaOH(aq) by the reaction
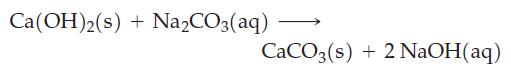
Transcribed Image Text:
Ca(OH)2(s) + Na₂CO3(aq) CaCO3(s) + 2NaOH(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
CaOH2s Na2CO3aq CaCO3s 2 NaOHaq Show that in principle Na2CO3aq can be converted almost completely t...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
Formation of the acid chloride will be done using a 100-mL round-bottom flask (RBF) with stir bar. Make sure that the glassware used is dry. Stick a wad of glass wool on the top of the condenser to...
-
Find any horizontal or vertical asymptotes. f(x) = = X 1- X
-
Walsh Company purchased $65,000, 10%, three-year bonds for $68,407.39. Interest on the bonds is payable semiannually. Using straight-line amortization of the bond premiums, make the necessary journal...
-
Create a stored procedure named prc_new_rental to insert new rows in the RENTAL table. The procedure should satisfy the following conditions. a. The membership number will be provided as a parameter....
-
Stone Brewing Co. is a San Diego brewer that has sold its beers for over two decades. Stone has maintained its trademark and brand from the beginning, registering the STONE mark in 1998. Stone has...
-
The Everly Equipment Company purchased a machine 5 years ago at a cost of $90,000. The machine had an expected life of 10 years at the time of purchase, and it is being depreciated by the...
-
3. Market demand is given by: 100-Q, P(Q) = if Q <100 otherwise Suppose there are two firms both with constant marginal cost 50 and no fixed cost and capacity constraint. Assume that firms produce...
-
Would you expect the lattice energy of MgS(s) to be less than, greater than, or about the same as that of MgO(s)? Use appropriate data from various locations in this text to obtain the values of the...
-
The reaction of borax, calcium fluoride, and concentrated sulfuric acid yields sodium hydrogen sulfate, calcium sulfate, water, and boron trifluoride as products. Write a balanced equation for this...
-
Water at 70F flows by gravity from a large reservoir at a high elevation to a smaller one through a 60-ft-long, 2-in-diameter cast iron piping system that includes four standard flanged elbows, a...
-
2 Solve w = 49, where w is a real number. Simplify your answer as much as possible. If there is more than one solution, separate them with commas. If there is no solution, click on "No solution". 2...
-
Find all values of x for the given function where the tangent line is horizontal. f(x)=x+12x+46
-
Your overseas forwarding agent advises you that he has received bookings (the merchandise is ready to ship) for the following merchandise: Factory 1 - 200 Cartons - 25 cbms - Customer Kohl's Factory...
-
2.) A 5 kg cat on a 2 kg skateboard starts at rest at the top of a 3 m tall ramp. The cat goes down the ramp, then across a rough horizontal surface 2 m long. The friction between the skateboard and...
-
In the H NMR spectrum of 2a, how many signals do you expect to appear in the range 3-4 ppm, and how many signals in the range 5-6 ppm? O Four signals in the 3-4 ppm range, and three signals in the...
-
Describe the three requirements for a valid offer.
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
These bonds were rated AA- by S&P. Would you consider them investment-grade or junk bonds?
-
What factors determine a companys bond rating?
-
Suppose you owned a portfolio consisting of $250,000 of long-term U.S. government bonds. a. Would your portfolio be riskless? Explain. b. Now suppose the portfolio consists of $250,000 of 30-day...
-
Gamze and Warda Confectioner Company produces two types of product; "chocolate" and "candy". At the end of 2023, a production plan related to these two products is being made. According to this plan,...
-
Prepare Trading and Profit and Loss account from the following information for the year 31st December 2004. of Opening stock 5,500 DYPurchases 18,400 Manufacturing wages 4,400 Sales 32,000 DY...
-
Desmond Co. is considering the following alternative financing plans: Plan 1 Plan 2 Issue 10% bonds (at face value) $1,080,000 $540,000 Issue preferred $1 stock, $10 par 900,000 Issue common stock,...

Study smarter with the SolutionInn App


