The situation pictured in Figure 6-10(a) is changed to that in Figure 6-10(b). What is the gas
Question:
The situation pictured in Figure 6-10(a) is changed to that in Figure 6-10(b). What is the gas pressure in Figure 6-10(b)?
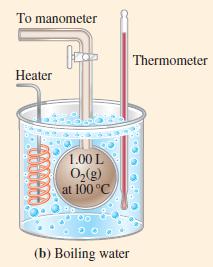
Figure 6-10(a)
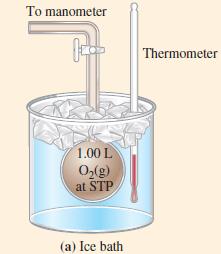
Figure 6-10(b)
Transcribed Image Text:
To manometer 1.00 L O₂(g) at STP (a) Ice bath Thermometer
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Analyze Identify the quantities in the general gas equation that remai...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2551+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
In the situation pictured in question 3, if there is a frictional force opposing the motion of the block, does this frictional force do work on the block? Explain.
-
In the situation pictured in question 28, is the magnitude of the velocity important to the success of the shot? Explain. Data from problem 28 A Q28 Diagram
-
A jet is traveling westward with the sun directly overhead (the jet is on a line between the sun and the center of the Earth). How fast must the jet fly in order to keep the sun directly overhead?...
-
What are the four perspectives of the balanced scorecard?
-
Given the stresses relative to compass coordinates (x = east, y = north, z = up), xx = 2,155, yy = 3,045, zz = 4,200, yx = 1,222, xz = 0, yx = 0 in psi with compression positive, find the principal...
-
The balance sheet of Banc One Corp. at December 31, 2007, reported 500,000 shares of \(\$ 1\) par common stock authorized with 100,000 shares issued. Paid-In Capital in Excess of Par had a balance of...
-
Patel Service Co. does make a few sales on account but is mostly a cash business. Consequently, it uses the direct write-off method to account for uncollectible accounts. During 2016 Patel Service...
-
The Garcia Company s bonds have a face value of $ 1 , 0 0 0 , will mature in 1 0 years, and carry a coupon rate of 1 6 percent. Assume interest payments are made semiannually. a . Determine the...
-
Without performing an actual calculation, indicate which has the greater u rms , He(g) at 1000 K or H 2 (g) at 250 K.
-
(A) A 1.00 mL sample of N 2 (g) at 36.2 C and 2.14 atm is heated to 37.8 C, and the pressure changed to 1.02 atm. What volume does the gas occupy at this final temperature and pressure? (B) Suppose...
-
What is variable costing?
-
Acting as the HIPAA Privacy Officer, identify the non-compliant issues in the following scenario and recommend corrective actions Background: The HIPAA Security Rule specifies that routine audits and...
-
IP Addressing: IP addressing will have significant impacts on the future extension of the network. You need to decide whether you will be using IPv4 or IPv6. Whatever you are choosing, you need to...
-
How could a hospital firm use the mix variance to analyze its revenues? How could a hospital firm use the mix variances to analyze salary costs regarding emergency room services?
-
Throughout American history, there has been consistent debate over the US Supreme Court's role in our political system and how it ought to make its decisions on cases. What do you think the US...
-
You have been tasked with introducing a new security policy to your campus. The new policy requires that all students and employees wear identification badges with their name and picture and that...
-
Consider a perfectly insulated and sealed container. Determine the minimum volume of a container such that a gallon of water at 25oC will evaporate completely. If the container is a cube, determine...
-
Consider a game of poker being played with a standard 52-card deck (four suits, each of which has 13 different denominations of cards). At a certain point in the game, six cards have been exposed. Of...
-
(Two Temporary Differences, Multiple Rates, Future Taxable Income) Flynn Inc. has two temporary differences at the end of 2010. The first difference stems from installment sales, and the second one...
-
(Two Differences, One Rate, First Year) The differences between the book basis and tax basis of the assets and liabilities of Morgan Corporation at the end of 2010 are presented below. It is...
-
(NOL Carry back and Carry forward, Valuation Account versus No Valuation Account) Sondgeroth Inc. reports the following pretax income (loss) for both financial reporting purposes and tax purposes....
-
Based on Sustainability in IKEA Group's case, how should IKEA deal with "sustainability cost" without disturbing its customer value (i.e., quality and price)?
-
Q.1) Mayur Batteries Ltd offered to public 16000 equity shares of Rs 10 each at a premium of Rs 2 per share. The amount payable is as follows: On application Rs 2 per share On allotment Rs 5 per...
-
Assume that the John Henry Company had $ 1 , 0 0 0 , 0 0 0 of sales during each of 2 0 2 2 and 2 0 2 3 , and it purchased merchandise costing $ 5 0 0 , 0 0 0 during each of those years. Its beginning...

Study smarter with the SolutionInn App


