The standard enthalpy of formation of H 2 O 2 (g) is -136 kJ mol -1 .
Question:
The standard enthalpy of formation of H2O2(g) is -136 kJ mol-1. Use this value, with other appropriate data from the text, to estimate the oxygen-to-oxygen single-bond energy. Compare your result with the value listed in Table 10.3.
Table 10.3
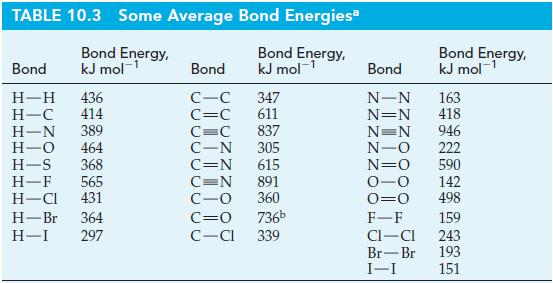
Transcribed Image Text:
TABLE 10.3 Some Average Bond Energies Bond Energy, kJ mol-¹ Bond Energy, kJ mol-¹ Bond H-H 436 H-C 414 H-N 389 H-O 464 H-S 368 H-F 565 H-Cl 431 H-Br 364 H-I 297 Bond C-C C=C 347 611 837 305 C=C C-N C=N 615 C=N 891 C-O 360 C=O 736b C-CI 339 Bond Energy, kJ mol¹ Bond N-N 163 N=N 418 N=N 946 N-O 222 N=O 590 0-0 142 0=0 498 F-F 159 CI-CI 243 Br-Br 193 I-I 151
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
HOs 12028 HO8 AHAH bonds broken AH bonds formed AH H0H0120...View the full answer

Answered By

Jeff Omollo
As an educator I have had the opportunity to work with students of all ages and backgrounds. Throughout my career, I have developed a teaching style that encourages student engagement and promotes active learning. My education and tutoring skills has enabled me to empower students to become lifelong learners.
5.00+
5+ Reviews
49+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Under Social Security, the family of a worker who dies while fully insured at the time of death has a right to survivors' benefits. True False
-
The head of a strike anywhere match contains tetraphosphorus trisulfide, P4S3. In an experiment, a student burned this compound in an excess of oxygen and found that it evolved 3651 kJ of heat per...
-
The standard enthalpy of formation of the metallocene bis (benzene) chromium was measured in a calorimeter. It was found for the reaction Cr (C6H6)2(s) Cr(s) + 2 C6H6 (g) that Uo (583 K) = +8.0 kJ...
-
1:When developing a marketing strategy for business customers, it is essential to understand the process the business goes through when making a buying decision. Knowledge of business buying behavior...
-
Joses Electronic Repair Shop has budgeted the following time and material for 2014. Joses budgets 5,000 hours of repair time in 2014 and will bill a profit of $5 per labor hour along with a 30%...
-
Convert the IP address whose hexadecimal representation is C22F1582 to dotted decimal notation.
-
Recommending Improvements to a Purchase Requisition Properly designed documents and forms improve the likelihood that employees will adhere to prescribed policies and procedures. For example,...
-
Abardeen Corporation borrowed $90,000 from the bank on October 1, 2016. The note had an 8percent annual rate of interest and matured on March 31, 2017. Interest and principal were paid in cash on the...
-
What is the angle in radians between vector r <4, 3> and vector s ?
-
Use the VSEPR theory to predict a probable shape of the molecule F 4 SCH 2 , and explain the source of any ambiguities in your prediction.
-
Estimate the enthalpy of formation of HCN using bond energies from Table 10.3, data from elsewhere in the text, and the reaction scheme outlined as follows. Table 10.3 C(g) A,H = ? HCN(g) AH = ? H(g)...
-
The J. R. Ryland Computer Company is considering a plant expansion that will enable the company to begin production of a new computer product. The companys president must determine whether to make...
-
The antibiotics erythromycin binds the 50S portion of the ribosome what impact will this drug have to a eukaryotic cell?
-
Question 6. Juicy Ltd produces and sells Fruit Juices. The company provides the following estimated sales for the next six months: Month January February March April May June Unit Sales 100,000...
-
Seventeen Seconds, Inc. is considering a long-term investment. The investment will require an investment of $40,000. It will have a useful life of 2 years, and no salvage (i.e., ending) value. Annual...
-
4. Given an 14-stage pipeline with branches resolved in the 10th stage. This processor can execute 4 instructions per cycle. (Show work) a. What is the CPI of the processor assuming 80% prediction...
-
Consider a 6-bit two's complement representation. Fill in the box with question mark "?" in the following table. You don't need to care about "n/a." Decimal Binary Number Representation...
-
Refer to the data in Exercise E24- 17. Calculate each divisions RI. Interpret your results. Exercise E24-17 Net Sales Residential 520,000 Professiona ,020,000 Operating Income 64,320 158,760 Average...
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
Accounting for a byproduct. Washington Oceanic Water (WOW) desalinates and bottles sea water. The desalinated water is in high demand from a large group of environmentally conscious people on the...
-
Alternative methods of joint-cost allocation, product-mix decisions. The Sunshine Oil Company buys crude vegetable oil. Refining this oil results in four products at the splitoft point; A, B, C, and...
-
Comparison of alternative joint-cost-allocation methods, further-processing decision, chocolate products. The Chocolate Factory manufactures and distributes chocolate products. It purchases cocoa...
-
4. A ball of mass 0.25 kg is thrown vertically upward from the roof of a building 18 m high with a speed of 16 m/s, and just misses the building on the way down, as shown. [3 marks] With what...
-
5. A 2.0 kg trolley moving east at 3.0 m/s collides head-on with a 1.0 kg trolley moving west at 2.0 m/s.. After the collision, the 2.0 kg trolley has a velocity of 1.0 m/s [E]. [4 marks] (a) What is...
-
A 2.0 kg mass is placed against a spring of force constant 800 N/m, which has been compressed 0.22 m, as shown. The spring is released, and the object moves along the horizontal, frictionless surface...

Study smarter with the SolutionInn App


