The structure of the molecule allene, CH 2 CCH 2 , is shown here. Propose hybridization schemes
Question:
The structure of the molecule allene, CH2CCH2, is shown here. Propose hybridization schemes for the C atoms in this molecule.
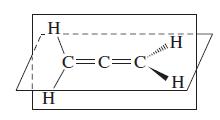
Transcribed Image Text:
H H C=C=C H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Based on the image you provided the hybridization schemes for the C atoms in allene are as fo...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Indicate which of the following molecules and ions are linear, which are planar, and which are neither. Then propose hybridization schemes for the central atoms. (a) Cl 2 C = CCl 2 ; (b) N C C N;...
-
The Lewis structure for allene is Make a sketch of the structure of this molecule that is analogous to Figure 9.25. In addition, answer the following three questions: (a) Is the molecule planar? (b)...
-
Here are shown the atomic packing schemes for several different crystallographic directions for some hypothetical metal. For each direction the circles represent only those atoms contained within a...
-
A horizontal jet of water (at 10C) that is 6 cm in diameter and has a velocity of 20 m/s is deflected by the vane as shown. If the vane is moving at a rate of 7 m/s in the x-direction, what...
-
Georgia purchased an option on Greenacre from Pamela for $10,000. The option contract contained a provision by which Georgia promised not to assign the option contract without Pamelas permission....
-
Determine F12 and F21 for the following configurations using the reciprocity theorem and other basic shape factor relations. Do not use tables or charts. (a) Long duct (b) Small sphere of area A1...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 334A? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
Gordon is the only limited partner in Bushmill Ventures, a limited partnership whose general partners are Daniels and McKenna. Gordon contributed $10,000 for his limited partnership interest and...
-
If $300,000 is to be saved over 15 years, how much should be deposited monthly if the investment earns 7% interest compounded monthly?
-
Angelic acid, shown below, occurs in sumbol root, a herb used as a stimulant. Represent the bonding in the angelic acid molecule by using the method in Figure 11-19 to indicate hybridization schemes...
-
Propose a bonding scheme that is consistent with the structure for propynal. 123 -C- -C 120 H -C- 1/7/7/196 120.4 pm 106 pm 146 pm 108 pm H 121 pm
-
Thomas is single and a self-employed architect. During 2021, Thomass income from his business is $170,000. He also pays $2,200 for a medical insurance policy. a. How should the medical insurance...
-
Explain what classes of stock Carter's Inc has, the characteristics of each class of stock, and the par value of the common stock. Explain how many common shares are authorized and issued and...
-
Can anyone in the class explain what a sale/leaseback arrangement is andwhy a managerial accountant would be interested in such an arrangement? What would be the advantages and disadvantages of this...
-
What are the recipients of the work expected to abide by, according to the copyright notice?
-
Define the accrued liabilities related to accrued commissions. write a brief intro on accured liabilities.
-
Compare and contrast long-form and short-form reports. Consider the following in your response: Explain when each report type should be used. Explain the purpose of the components that should be...
-
At the beginning of 2011, the Healthy Life Food Company purchased equipment for $42 million to be used in the manufacture of a new line of gourmet frozen foods. The equipment was estimated to have a...
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
Operating Cycle what are some of the characteristics of a firm with a long operating cycle?
-
Cash Cycle what are some of the characteristics of a firm with a long cash cycle?
-
Sources and Uses for the year just ended, you have gathered the following information about the Holly Corporation. a. A $200 dividend was paid. b. Accounts payable increased by $500. c. Fixed asset...
-
Choose an organization such as Samsung, Starbucks, Ford Motor Company, or Waste Management that implemented a major change. For example, a sustainability initiative at Starbucks or Apple making...
-
1. Law enforcement is primarily the responsibility of local government (77% of the nation's police employees work at the local level). Why do we not have or want a national police force and do you...
-
Why did Marcy develop breasts? Marcys testes were removed because a malignancy can develop in them. Why did she require estrogen replacement therapy after the surgery? Why did George have a poor...

Study smarter with the SolutionInn App


