Two of the substances listed here are highly soluble in water, two are only slightly soluble in
Question:
Two of the substances listed here are highly soluble in water, two are only slightly soluble in water, and two are insoluble in water. Indicate the situation you expect for each one.
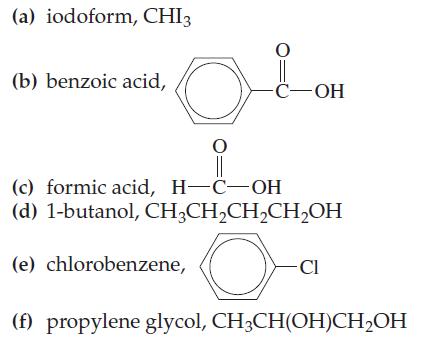
Transcribed Image Text:
(a) iodoform, CHI3 (b) benzoic acid, O O || (c) formic acid, H-C-OH (d) 1-butanol, CH₂CH₂CH₂CH₂OH O (e) chlorobenzene, -C-OH - CI (f) propylene glycol, CH3CH(OH)CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Substance Solubility Explanation iodoform CHI3 Highly soluble Iodoform is a nonpolar molecule but it ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Benzoic acid, C 6 H 5 COOH, is much more soluble in NaOH(aq) than it is in pure water. Can you suggest a reason for this? The structural formula for benzoic acid is given in Exercise 5(b). Exercise...
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
A sealed flask contains water and oxygen gas at 25C. The O 2 gas has a partial pressure of 1.5 atm. (a) What is the concentration of O 2 in the water? (b) If the pressure of O 2 in the flask is...
-
Green Pastures is a 400-acre farm on the outskirts of the Kentucky Bluegrass, specializing in the boarding of broodmares and their foals. A recent economic downturn in the thoroughbred industry has...
-
A beam of laser light of wavelength 632.8 nm has a circular cross section 2.00 mm in diameter. A rectangular aperture is to be placed in the center of the beam so that, when the light falls...
-
Compare and contrast between various valuation methods. Which valuation methods are most appropriate for manufacturing firms and which are the most appropriate for internet-based ventures?
-
Lisa Company had 100 units in beginning inventory at a total cost of $10,000. The company purchased 200 units at a total cost of $26,000. At the end of the year, Lisa had 80units in ending inventory...
-
Let's dive into the IRAC method, a fundamental framework in legal analysis. Each part, Issue, Rule, Analysis, and Conclusion, plays a unique role. Think about which part might be most crucial for a...
-
At 0 C and an O 2 pressure of 1.00 atm, the aqueous solubility of O 2 (g) is 48.9 mL O 2 per liter. What is the molarity of O 2 in a saturated water solution when the O 2 is under its normal partial...
-
Describe a case in which the liquid and vapor curves in a diagram such as Figure 14-16 would converge into a single curve. Is such a case likely to exist? Figure 14-16 Pressure, mmHg 100.0 90.0 80.0...
-
Kathryn is a waitress at a restaurant in South Carolina. She earns the tipped minimum wage. During a 40-hour work week, how much must she earn in tips to satisfy the minimum wage requirement? 1....
-
find a current events article related to a macroeconomic topic (unemployment, inflation, economic growth, taxes, etc.) that summarizes the article, assess the article (what did you learn? Do you...
-
Describe one take-away from quantitative, qualitative, and mixed method research. Also, how will you analyze and use research in the future to help improve your organization?
-
The dependent variable in my study is employee performance and the independent variable is leadership. the Moderator Variable variables are Perceived Organizational Support (POS) and Emotional...
-
Provide your recommendation in regard to the investment in Microsoft stock. -You should include a qualitative analysis where you detail the reasons supporting your chosen "super-normal growth rate"....
-
A leading beverage company sells its signature soft drink brand in vending machines for $0.89 per 12 oz. can. A vending machine has monthly costs of space rental, energy consumption, and capital...
-
What is the relationship between managements financial statement assertions and audit evidence?
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
In each case, state which of the two securities is likely to provide the higher return: (a) When the stock price rises (stock or convertible bond?). (b) When interest rates fall (straight bond or...
-
In 1996 Marriott International made an issue of LYONS. The bond matured in 2011, had a zero coupon, and was issued at $532.15. It could be converted into 8.76 shares. Beginning in 1999 the bonds...
-
The companys decision to issue warrants should depend on the managements forecast of likely returns on the stock. Do you agree?
-
What three steps would you put in place to ensure that YOUR brand and its advertising campaigns are approved before they go to print or tv?
-
Marketing plan to support your customer's efforts in using/carrying your product (Light Bulb with CCTV) Please give an example and explanation
-
Maroon Inc. currently owes a balance of $110,694.10 on a $60,000.00 loan. If the loan has an interest rate of 11.50% compounded monthly. How long has the loan been outstanding?

Study smarter with the SolutionInn App


