Use 1 x 10 -13 cm as the approximate diameter of the spherical nucleus of the hydrogen-1
Question:
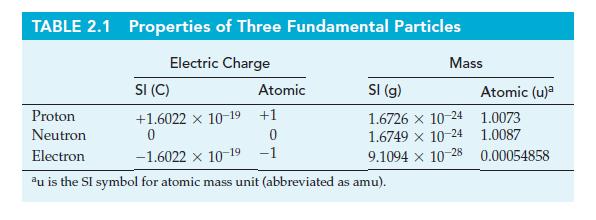
Use 1 x 10-13 cm as the approximate diameter of the spherical nucleus of the hydrogen-1 atom, together with data from Table 2.1, to estimate the density of matter in a proton.
Table 2.1
Transcribed Image Text:
TABLE 2.1 Properties of Three Fundamental Particles Electric Charge Atomic Mass SI (C) Proton +1.6022 x 10-19 +1 Neutron 0 0 Electron -1.6022 × 10-19 -1 au is the SI symbol for atomic mass unit (abbreviated as amu). SI (g) 1.6726 x 10-24 1.6749 × 10-24 9.1094 x 10-28 Atomic (u)² 1.0073 1.0087 0.00054858
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Density d massg volume gcm mass g proton 16726x1...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The hydrogen atom is composed of one proton in the nucleus and one electron, which moves about the nucleus. In the quantum theory of atomic structure, it is assumed that the electron does not move in...
-
The large magnetic fields used in MRI can produce forces on electric currents within the human body. This effect has been proposed as a possible method for imaging biocurrents flowing in the body,...
-
An atom has a very small nucleus surrounded by an electron cloud. Figure 2.1 represents the nucleus with a diameter of about 2 mm and describes the electron cloud as extending over 200 m. If the...
-
Each of the following passages may be plausibly criticized by some who conclude that it contains a fallacy, but each may be defended by some who deny that the argument is fallacious. Discuss the...
-
a. A vacant lot acquired for $75,000 is sold for $145,000 in cash. What is the effect of the sale on the total amount of the sellers? (1) Assets (2) Liabilities (3) Owner's equity? b. Assume that the...
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. All agreements between two competent parties are contracts.
-
T. Christian Cooper was a partner to Sanders and Richard Campbell d/b/a The Mullen Company. In 2001, Cooper helped bring about a management agreement between The Mullen Co. and Newnan Crossing...
-
The Carlson Department Store suffered heavy damage when a hurricane struck on August 31, 2003. The store was closed for four months (September 2003 through December 2003), and Carlson is now involved...
-
1. You invest $2000 in an account that pays simple interest of 7% for 10 years. The amount of money you'll have at the end of 10 years is: 2. Use the compound interest formula to determine the...
-
Fluorine has a single atomic species, 19 F. Determine the atomic mass of 19 F. by summing the masses of its protons, neutrons, and electrons, and compare your results with the value listed on the...
-
William Prout (1815) proposed that all other atoms are built up of hydrogen atoms, suggesting that all elements should have integral atomic masses based on an atomic mass of one for hydrogen. This...
-
The Lavalle Ladies Legends (LLL), a professional hockey team playing in the Canadian Hockey League, is preparing an outline forecast of its pro forma income statement for 2010. The president of the...
-
Planning Real Estate Office CSR Event Customer complaints are up 25% in the last 12-month period Data collected from surveys, tickets tracked through Customer Support (email, letter, and phone...
-
For Final Research Paper, you will create a comprehensive study defining your personal choice of the personality theory you identified in the Week Three assignment. In a six- to eight-page paper,...
-
Using the policies and procedures for Gold Catering: Explain the different procedures involved in recording bad debts and doubtful debts: Why is there a difference between bad debt and doubtful debt?...
-
What is firmware? How are computer viruses different from worms? Explain briefly
-
Describe the fundamental principles of electricity generation. Explain the relationship between electrical current and magnetism. Finally, provide examples where these apply to the operation of an...
-
Shrute Inc. manufactures office copiers, which are sold to retailers. The price and cost of goods sold for each copier are as follows: Price ..............$ 1,110 per unit Cost of goods sold ..........
-
Why is it important to understand the macro-environment when making decisions about an international retail venture?
-
What are two potential tests that can be conducted to verify the CAPM? What are the results of such tests? What is rolls critique of CAPM tests?
-
Briefly explain the difference between the CAPM and the arbitrage pricing theory (APT).
-
Suppose you are given the following information. The beta of company, bi, is 0.9, the risk free rate, rRF, is 6.8%, and the expected market premium, rM-rRF, is 6.3%. Because your company is larger...
-
Problem 5 Suppose the 12-month forward price of the pound in terms of dollars is 1.5 dollars per pound and the spot price of of the pound in terms of dollars is 1.55. Assume also that currently the...
-
price equilibrium of studio apartment is 2K, and unit equilibrium is 5K, FED pass out a new law that saying 1.2K is price for 1 studio apartment. Draw supply and demand graph, (price ceiling and...
-
Banking Question 2 Please use the balance sheet for Bank Alpha to answer the questions. Assume Bank Alpha loans all of its excess reserves and the assets and liabilities balance on the balance sheet....

Study smarter with the SolutionInn App


