Use data from Table 2.1 to verify that (a) The mass of electrons is about 1/2000 that
Question:
Use data from Table 2.1 to verify that
(a) The mass of electrons is about 1/2000 that of H atoms;
(b) The mass-to-charge ratio (m/e) for positive ions is considerably larger than that for electrons.
Table 2.1
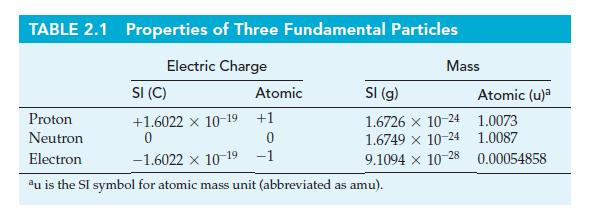
Transcribed Image Text:
TABLE 2.1 Properties of Three Fundamental Particles Electric Charge SI (C) +1.6022 x 10-19 +1 0 Atomic Mass SI (g) 1.6726 x 10-24 1.6749 x 10-24 9.1094 x 10-28 Proton Neutron 0 Electron -1.6022 × 10-19 -1 au is the SI symbol for atomic mass unit (abbreviated as amu). Atomic (u)a 1.0073 1.0087 0.00054858
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a Verify that the mass of ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use data from Table to estimate ÎH for the combustion of methane (CH4), as shown below: Table s | 14 39 95 45 72 1 1419 6847064968 77386 42222 34 985 0302 121 Si H C O 437 490 9 31222241122...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Which of the following statements is correct regarding a work sheet and the adjustment process? Adjusting journal entries are prepared from the Adjusted Trial Balance columns. Adjusting journal...
-
Wiley, Inc., an MNC, has a beta of 1.3. The U.S. stock market is expected to generate an annual return of 11 percent. Currently, Treasury bonds yield 2 percent. Based on this information, what is...
-
Melanie Mielke Construction Corporation is considering the appropriate accounting for two unrelated events during the year. The first event related to the effects of a labor strike that resulted in a...
-
99 percent per month. In 2014, Boling resolved his suit against the gas can manufacturer. Shortly thereafter, Prospect sent Boling a Schedule of Purchases, asserting that Boling owed Prospect...
-
The ledger of American Company has the following work in process account. Production records show that there were 400 units in the beginning inventory, 30% complete, 1,600 units started, and 1,700...
-
Prof. Dimitriou developed the following code to evaluate the expressions A(), B() and CO) but it takes too long to compute even for small values of n. A(int n) if (n <0) return 0; if (n
-
Determine the approximate value of m/e in grams per coulomb for the ions 127 53 I - and 32 16 S 2 - Why are these values only approximate?
-
The following observations were made for a series of five oil drops in an experiment similar to Millikans (see Figure 2-8). Drop 1 carried a charge of 1.28 x 10 -18 C; drops 2 and 3 each carried 1 /...
-
For each of the following task environment properties, rank the example task environments from most to least according to how well the environment satisfies the property. Lay out any assumptions you...
-
M-E Drafting Incorporated reports the following statement of stockholders' equity. Solve for the missing amounts. Note: Amounts to be deducted should be indicated by a minus sign. Financial...
-
1. Let set U= (n | ne whole numbers, 10
-
Consider the following ad hoc network with all bi-directional links. Suppose node A needs to find a route to node E using DSR (Dynamic Source Routing) using route caching and optimizations. Assume...
-
A network administrator is setting up a web-based application for a company. The application needs to be continually accessible to all end users. Which of the following would best ensure this need is...
-
A technician is configuring a wireless access point in a public space for guests to use. Which of the following should the technician configure so that only approved connections are allowed? A....
-
From the following accounts (not in order), prepare a post-closing trial balance for Winter Co. on October 31, 201X. $19,950 9,700 P. Winter, Capital P. Winter, Withdrawals Accounts Receivable Legal...
-
What are current assets and current liabilities? How are they different from non-current assets and non-current liabilities?
-
Do the following events represent business transactions? Explain your answer in each case. (a) A computer is purchased on account. (b) A customer returns merchandise and is given credit on account....
-
Name the accounts debited and credited for each of the following transactions. (a) Billing a customer for work done. (b) Receipt of cash from customer on account. (c) Purchase of office supplies on...
-
Why are revenue and expense accounts called temporary or nominal accounts?
-
The magnitude of the electric field at a point P is 2.3 x 10^7 NIC. What is the magnitude of the electric force that would be exerted at P on (a) an electron? (b) an oxygen nucleus (charge +8e)?
-
A 0.0110 kg bullet is fired at v 0=554 m/s into a solid cylinder of mass 17.1 kg and radius 0.290 m (moment of inertia: I=1/2 mR 2). The cylinder is initially at rest and is mounted on the fixed...

Study smarter with the SolutionInn App


