Use data from Table 7.2 to calculate the volume of butane, C 4 H 10 (g), measured
Question:
Use data from Table 7.2 to calculate the volume of butane, C4H10(g), measured at 24.6 °C and 756 mmHg, that must be burned to liberate 5.00 x 104 kJ of heat.
Table 7.2
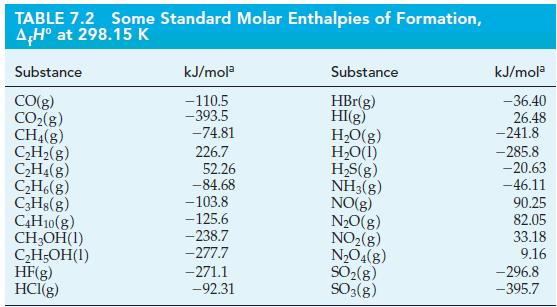
Transcribed Image Text:
TABLE 7.2 Some Standard Molar Enthalpies of Formation, AH° at 298.15 K Substance CO(g) CO₂(g) CH4(8) C₂H₂(g) C₂H4(g) C₂H6(8) C3H8(g) C4H10(g) CH₂OH(1) C₂H5OH(1) HF(g) HCI(g) kJ/mola -110.5 -393.5 -74.81 226.7 52.26 -84.68 -103.8 -125.6 -238.7 -277.7 -271.1 -92.31 Substance HBr(g) HI(g) H₂O(g) H₂O(1) H₂S(g) NH3(g) NO(g) N₂O(g) NO₂(g) N₂O4(g) SO₂(g) SO3(g) kJ/mola -36.40 26.48 -241.8 -285.8 -20.63 -46.11 90.25 82.05 33.18 9.16 -296.8 -395.7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To calculate the amount of butane C4H10 that must be burned to liberate 500 104 kJ of heat we need t...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Calculate the mass of octane, C8H18(l), that must be burned in air to evolve the same quantity of energy as produced by the fusion of 1.0 g of hydrogen in the following fusion reaction: Assume that...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Carol Harris, Ph.D, CPA, is a single taxpayer and she lives at 674 Yankee Street, Durham, NC 27409. Her Social Security number is 793-52-4335. Carol is an Associate Professor of Accounting at a local...
-
is an acute angle such that tan() =1/3. What is the value of 1010. (sin + cos)? 3.
-
Fast Ink produces ink-jet printers for personal computers. It received an order for 400 printers from a customer. The following information is available for this order. Process time . . . . . . . . ....
-
Determine the stresses in all parts of the bar shown in Fig. 13.60 and rigidly clamped at the ends. A B 10mm 80kN 60kN 400mm -200mm -300mm- Fig. 13.60
-
Do consumer products and services perform missions?
-
The records of Burris Inc. reflect the following data: Work in process, beginning of month-2,000 units one-half completed at a cost of $1,250 for materials, $675 for labor, and $950 for overhead....
-
Quilcene Oysteria Flexible Budget For the Month Ended August 31 Actual pounds (q) 7,100 Revenue ($4.05q) $ 28,755 Expenses: Packing supplies ($0.25q) 1,775 Oyster bed maintenance ($3,400) 3,400 Wages...
-
Categorize each of the following characteristics as being more representative of either traditional manufacturing or lean production. 1. Quality tends to be inspected-in rather than built-in. 2....
-
The decomposition of limestone, CaCO 3 (s), into quicklime, CaO(s), and CO 2 (g) is carried out in a gas-fired kiln. Use data from Appendix D to determine how much heat is required to decompose 1.35...
-
Use data from Table 7.3 and Appendix D to determine r H the following reaction. Table 7.3 Mg(OH)2(s) + 2NH4+ (aq) Mg2+ (aq) + 2 HO(1) + 2NH3(g) AH = ?
-
Reggie is a self-employed taxpayer who turns 59 years old at the end of the year (2014). In2014, his net Schedule C income was $300,000. This was his only source of income. This year, Reggie is...
-
Problem 3-4 (Past exam question: 40 minutes) (Text: Ch. 3) Graham Grepzer has come to you in connection with two offers of employment as a salesperson from two rival dry-goods manufacturing...
-
Dale, a 40 year old single taxpayer, incurs medical expenses for the year as follows: Medical insurance premiums: $11,000 Doctors and hospital fees: $3,500 Hearing aids : $ 350 General purpose...
-
David Harbert recently started a business. During the first few days of operation, Mr. Harbert transferred cash from his personal account into a business account for a company he named Harbert...
-
1. Define 5D in your own words. If it was defined as an operation that uses "trochoidal passes with a big and tightly controlled maximal radial engagement", what operation is being referred to? 2. Is...
-
Question 3. Prove by induction the identity (n+1)(n+2)... (2n-1) 2n 1.3.5.... (2n-1) for any natural n. 2n
-
A management consulting company presents a three-day seminar on project management to various clients. The seminar is basically the same each time it is given. However, sometimes it is presented to...
-
In order to get an idea on current buying trends, a real estate agent collects data on 10 recent house sales in the area. Specifically, she notes the number of bedrooms in each house as follows: a....
-
Purchase of Computer with Zero-Interest-Bearing Debt Napoleon Corporation purchased a computer on December 31, 2009, for $130,000, paying $30,000 down and agreeing to pay the balance in five equal...
-
Asset Acquisition Logan Industries purchased the following assets and constructed a building as well. All this was done during the current year. Assets 1 and 2 These assets were purchased as a lump...
-
Nonmonetary Exchange Alatorre Corporation, which manufactures shoes, hired a recent college graduate to work in its accounting department. On the first day of work, the accountant was assigned to...
-
Baird Manufacturing Company established the following standard price and cost data: Sales price Variable manufacturing cost Fixed manufacturing cost Fixed selling and administrative cost $ 8.80 per...
-
The management of Zigby Manufacturing prepared the following balance sheet for March 31. Cash Assets ZIGBY MANUFACTURING Balance Sheet March 31 Liabilities Liabilities and Equity $ 48,000 Accounts...
-
Sonic Wave Corporation's plantwide predetermined overhead rate is $16.00 per direct labor-hour and its direct labor wage rate is $20.00 per hour. The following information pertains to Job B-300. What...

Study smarter with the SolutionInn App


