Use the data in Appendix D to calculate the standard cell potential for each of the following
Question:
Use the data in Appendix D to calculate the standard cell potential for each of the following reactions. Which reactions will occur spontaneously?
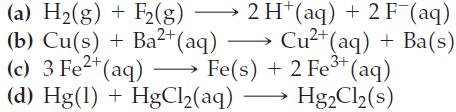
Transcribed Image Text:
(a) H₂(g) + F2 (g) (b) Cu(s) + Ba²+ (aq) 2 H(aq) + 2 F (aq) Cu²+ (aq) + Ba(s) 3+ (c) 3 Fe²+ (aq) →→→→→→ Fe(s) + 2 Fe³+ (aq) (d) Hg(1) + HgCl₂(aq) Hg₂Cl₂ (s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To calculate the standard cell potential for each of the given reactions and determine which reactio...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use the data in Appendix D to calculate the standard cell potential for each of the following reactions. Which reactions will occur spontaneously? 2+ Fe2+ + (aq) + Ag+ (aq) 2 Sn+ (aq) Hg2 (a) Fe+...
-
1. List out Linear and Non Linear data structures and explain them briefly. 2a) Write an algorithm for Bubble Sort.Also sort the folowing numbers using Bubble sort: 45, 23, 5, 41,5, 1, 20 2b) Sort...
-
Use the data in Appendix 3 to calculate the equilibrium constant for the reaction Agl(s) Ag+(aq) + I2(aq) at 25C. Compare your result with the Ksp value in Table 16.2.
-
Question: If you were a consultant and for the below M&A case, what questions would you ask as a consultant of the acquiring company of the mine and in order to complete the valuation: A firm is...
-
What strategy might be employed to distribute goods effectively in the dichotomous small/large middleman pattern, which characterizes merchant middlemen in most countries?
-
Suppose that you have a number of capacitors. Each is identical to the capacitor that is already in a series RCL circuit. How many of these additional capacitors must be inserted in series in the...
-
Moore, Corp., completed the following selected transactions and prepared these adjusting entries during May: Requirements 1.State whether the transaction would increase revenues, decrease revenues,...
-
Internal Control Questionnaire Items: Assertions, Tests of Controls, and Possible Errors or Frauds. Following is a selection of items from the payroll processing internal control questionnaire in...
-
Why do firms make foreign direct investment (instead of domestic venturing and other modes of internationalisation)?Explain
-
Predict whether the following metals will react with the acid indicated. If a reaction does occur, write the net ionic equation for the reaction. Assume that reactants and products are in their...
-
Write cell reactions for the electrochemical cells diagrammed here, and use data from Table 19.1 to calculate E cell for each reaction. Table 19.1 2+ (a) Al(s) Al+ (aq)||Sn+ (aq)|Sn(s) (b) Pt(s) Fe2+...
-
Use spherical coordinates to find the volume of the solid. Solid inside x + y + z = 9, outside z = x + y and above the xy-plane
-
A small hair dresser shop operates with three hair stylists. It has two waiting seats so that when new customers arrive and all stylists are busy and there is no vacant seat, they leave. The state...
-
n A student wants to know how can she prove that a sequence whose nth term is 5n+4 is arithmetic and a sequence whose nth term is 5-3 is geometric. How do you respond? How can she prove that a...
-
Define a time class named HoursMinutes that will encapsulate time calculations. Store the time in a single private data member named totalMinutes. The class should provide the following methods: 1. a...
-
Organization Development - Reflections Reflection How do you feel about your experience? What did you learn and how did you benefit? What were the challenges you faced? Express your opinion about...
-
2. Fit auto-regressive (AR) models with regular and stochastic gradient de- scents. Refer to the two-tap predictor example in the slides of chapter 5a, consider a AR(2) model x(n) = -wx(n-1) - wx(n -...
-
On January 1, 2017, Vahsholtz Corporation purchased 5,000 shares of treasury stock. Other information regarding Vahsholtz Corporation is provided as follows. Compute (a) Return on common...
-
You are the newly appointed tax practitioner to complete Emilys tax return and have downloaded the prefill report for Emilys tax return (hint, you can read what a prefill report is here (Links to an...
-
In a recent quarter, Alliance Atlantis Communications Inc. reported cash provided by operating activities of $45,600,000 and revenues of $264,800,000. Cash spent on plant asset additions during the...
-
The management of Radar Inc. is trying to decide whether it can increase its dividend. During the current year it reported net income of $875,000. It had cash provided by operating activities of...
-
During the year, prepaid expenses decreased $6,600, and accrued expenses increased $2,400. Indicate how the changes in prepaid expenses and accrued expenses payable should be entered in the...
-
Cobalt Industries produces widgets. The 2023 budget and actuals were as follows: Budget Actual Production and sales (units) 6,000 5,400 Sales Revenue $ 1,380,000 $ 1,296,000 Variable costs $ 660,000...
-
Explain what is a "Balanced Scorecard" approach to performance management. Explain if this approach is suitable for designing a competency-based structure. Name four (4) key elements that were...
-
Lakeside Incorporated manufactures four lines of remote control boats and uses activity-based costing to calculate product cost. Activity Pools Machining Estimated Total Cost Setup $639,600 101,625...

Study smarter with the SolutionInn App


