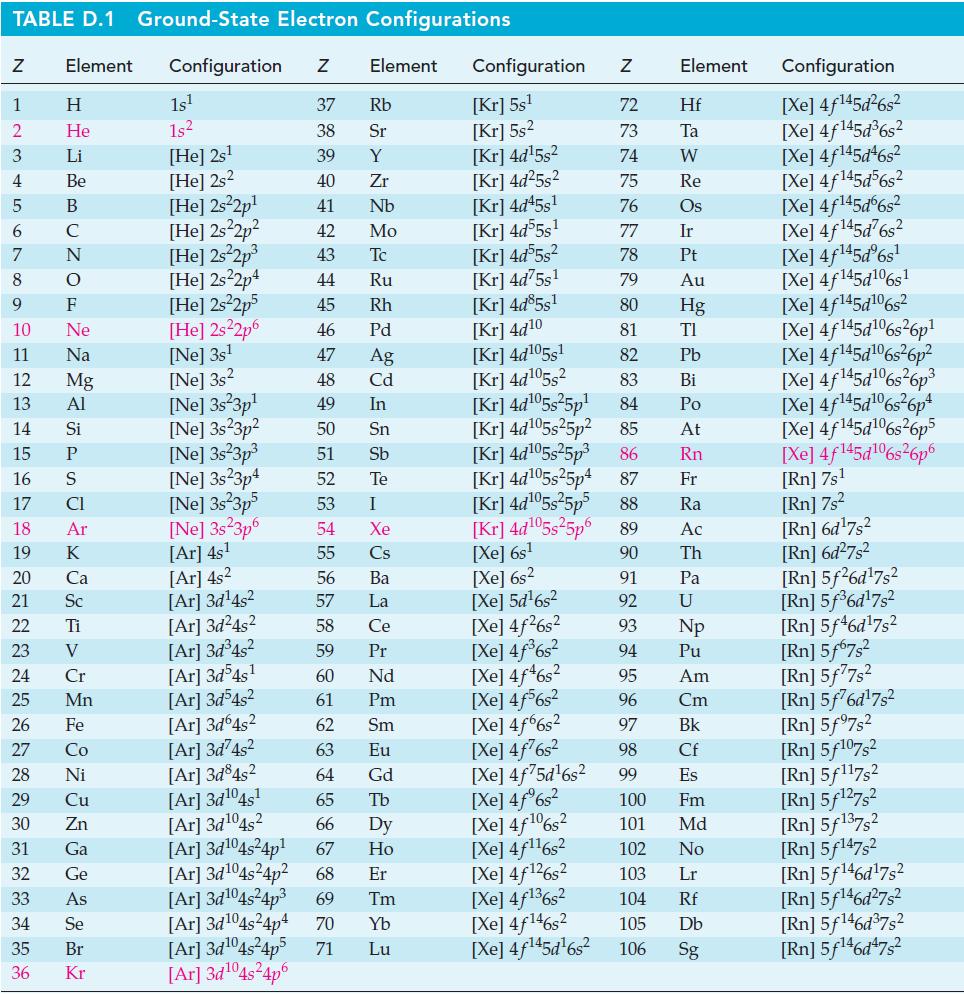
Use thermodynamic data from Appendix D to calculate a theoretical voltage of the silverzinc button cell described
Question:
Use thermodynamic data from Appendix D to calculate a theoretical voltage of the silver–zinc button cell described on page 893.

Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 HIG&LUZONSUZ SE> 0 ≤ 2 3 2 3 5 3 3 2 2 5 2 He 10 11 12 13 14 15 16 17 18 19 20 21 Sc 22 23 24 25 Mn Mg 26 27 28 Ni 29 30 31 32 33 34 35 36 Zn Ga Ge 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 54 55 [Ar] 4s² 56 [Ar]3d¹4s² 57 [Ar]3d²4s² 58 [Ar]3d³4s² 59 [Ar]3d54s¹ 60 61 [Ar]3d³4s² [Ar]3d64s² 62 [Ar]3d²4s² 63 [Ar]3d845² 64 [Ar]3d¹04s¹ 65 [Ar]3d¹04s2 [Ar]3d¹04s²4p¹ [Ar]3d¹04s²4p² [Ar]3d¹04s²4p³ 69 Tm 66 Dy 67 Ho 68 Er [Ar]3d¹04s²4p4 70 Yb Lu [Ar]3d¹04s²4p5 71 [Ar]3d¹04s²4p6 [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 Element [Ne] 3s 3p6 [Ar] 4s¹ I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd Tb Configuration Z [Kr] 5s¹ [Kr] 5s² [Kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [Kr] 4d55s¹ [Kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [Kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [Kr] 4d¹05s25p² [Kr] 4d¹05s²5p³ [Kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [Kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Lisper Wanja
I am an experienced and highly motivated writer with a passion for the skills listed. I have a proven track record of my expertise and my aim is to deliver quality, well-detailed and plagiarism free projects. My genuine passion for writing combined with my ongoing professional development through school and research makes me an ideal candidate within for any assignment.
4.90+
233+ Reviews
388+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The hydrazine fuel cell is based on the reaction The theoretical E cell of this fuel cell is 1.559 V. Use this information and data from Appendix D to calculate a value of f G for [N 2 H 4 (aq)]....
-
(A) Use electrode potential data to determine G for the reaction (B) The hydrogenoxygen fuel cell is a voltaic cell with a cell reaction of 2 H 2 (g) + O 2 (g) 2 H 2 O(l). Calculate E cell for this...
-
Use the Nernst equation and data from Appendix D to calculate E cell for each of the following cells. (a) Mn(s) Mn+ (0.40 M)||Cr+ (0.35 M), Cr+ (0.25 M) Pt(s) (b) Mg(s) Mg2+ (0.016 M)||[Al(OH)4](0.25...
-
In order to evaluate lim f(a+h)-f(), it is necessary to evaluate f(a + h). h xa For f(x) = x 3, f(a+h) =
-
Someone once commented that advertising is Americas greatest export. Discuss.
-
If a 15-cm-long column has a plate height of 5.0 m, what will be the half-width (in seconds) of a peak eluted at 10.0 min? If plate height = 25 m, what will be w1/2?
-
On December 31, 2018, Edmand, Inc., issued \(\$ 750,000\) of 11 percent, five-year bonds for \(\$ 722,400\), yielding an effective interest rate of 12 percent. Semiannual interest is payable on June...
-
A company that manufactures food and beverages in the vending industry has purchased some handling equipment that cost $75,000 and will be depreciated using MACRS GDS. The class life of the asset is...
-
Find 1 real life Canadian example within the last 10 years that have been documented in the news (newspaper, magazine, or video news) of breaches of the code of conduct. 1 needs to be an example from...
-
Determine the values of r G for the following reactions carried out in voltaic cells. (a) 2 Al(s) + 3 Cu+ (aq) (b) O(g) +41 (aq) + 4 H*(aq) 2 A1+ (aq) + 3 Cu(s) 2 HO(1) +2 12(s) (c) CrO7 (aq) + 14...
-
Write a cell diagram and calculate the value of E cell for a voltaic cell in which (a) Cl(g) is reduced to Cl(aq) and Fe(s) is oxidized to Fe+ (aq); (b) Ag (aq) is displaced from solution by Zn(s);...
-
The bookkeeper of a firm failed to agree the trial balance at 30 June, the end of the financial year. She opened a suspense account into which she entered the amount she was out of balance and...
-
Authorization of transactions is considered an important accounting control. The movement to computerized and paperless accounting systems poses new problems for management in implementing...
-
Why do organizations use groups to solve problems and make decisions?
-
A company involved in e-commerce would expect a firewall to do all of the following except: a. Intercept traffic that meets specific criteria and send the traffic back to the originator of the...
-
To obtain evidence that user identification and password control procedures are functioning as designed, an auditor would most likely a. Attempt to sign onto the system using invalid user...
-
Which of the following statements are correct regarding good control procedures over program change control? I. A programmer should be able to make changes to a program only while it is in the...
-
Graph these cost behaviour patterns over a relevant range of 010,000 units: a. Variable expenses of $6 per unit b. Mixed expenses made up of fixed costs of $30,000 and variable costs of $2 per unit...
-
What tools are available to help shoppers compare prices, features, and values and check other shoppers opinions?
-
Wilma Flintstone and Barny Rublestone were discussing the format of the statement of cash flows of Hart Candy Co. At the bottom of Hart Candys statement of cash flows was a separate section entitled...
-
Why is it necessary to use comparative balance sheets, a current income statement, and certain transaction data in preparing a statement of cash flows?
-
Contrast the advantages and disadvantages of the direct and indirect methods of preparing the statement of cash flows. Are both methods acceptable? Which method is preferred by the FASB? Which method...
-
Compare and contrast private equity and venture capital. What are the benefits of these kinds of financing approaches?
-
A bid may be disqualified in all the below situations, except: Question 8 options: When received after the closing date and time When the bid deposit amount does not match the amount requested When...
-
4 Suppose (|) is the posterior distribution for some parameter , and that it follows a Beta distribution with parameters and ; we will refer to these as shape parameters 1 and 2, to avoid confusion...

Study smarter with the SolutionInn App


