Various thermochemical cycles are being explored as possible sources of H 2 (g). The object is to
Question:
Various thermochemical cycles are being explored as possible sources of H2(g). The object is to find a series of reactions that can be conducted at moderate temperatures (about 500 °C) and that results in the decomposition of water into H2 and O2. Show that the following series of reactions meets these requirements.
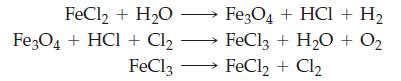
Transcribed Image Text:
FeCl₂ + H₂O Fe3O4 + HCl + Cl₂ Fe3O4 + HCl + H₂ FeCl3 + H₂O + 0₂ FeCl3 FeCl2 + Cl2 →→→
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To show that the given series of reactions meets the requirement of decomposing water into H2 and O2 ...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Show two different Friedel-Crafts acylation reactions that can be used to prepare the following compound. CH3 CH CH3
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Lucy has just been promoted to a managerial position and given a new office. She is very fond of small Persian carpets and Native American paintings and wants to get some carpets and paintings for...
-
At one time, Circle K was the second-largest convenience store chain in the United States. At its peak, Circle K operated 4,685 stores in 32 states. Circle Ks rapid expansion was financed through...
-
Many employees believe that their employers stock is less likely to lose half of its value than a well diversified portfolio of stocks. Explain why this belief is erroneous.
-
Johanna Marra and Eric Nazzaro began a romantic relationship in October 2013. That previous July, Nazzarro had purchased a duplex that he intended to renovate. Nazzarro rented out the top floor while...
-
Elsea Company, which produces and sells a small digital clock, bases its pricing strategy on a 25percent markup on total cost. Based on annual production costs for 25,000 units of product,...
-
Listed below are eight firms that are currently operating in the market. The market share of each firm is listed in the table below. Firm Market Share Bird 16 Cat 11 Dog 8 Horse 4 Pig 5 Cow 10 Tiger...
-
We have stressed the relationship between E values and thermodynamic properties. We can use this relationship to add some missing features to an electrode potential diagram. For example, note that...
-
Chemists have successfully synthesized the ionic compound [N 5 ][SbF 6 ], which consists of N 5 + and SbF 6 ions. Draw Lewis structures for these ions and assign formal charges to the atoms in your...
-
In a free-fall experiment, an object is dropped from a height of 256 feet. A camera on the ground 500 feet from the point of impact records the fall of the object (see figure). (a) Find the position...
-
Let \(\left(B_{t}ight)_{t \geqslant 0}\) be a \(\mathrm{BM}^{1}\). Show that \(\mathscr{F}_{\infty}^{B}=\bigcup_{J \text { countable }}^{J \subset[0, \infty)} \sigma(B(t): t \in J)\).
-
A 2016 survey by Opinion Way for kayak.co.uk found that the use of travel agents has changed dramatically over the past 10 years in the United Kingdom (Sullivan, 2016). While 57\% of respondents...
-
The iPhone debuted in January 2007. In the 10 years since then, smartphones have revolutionized and enriched our lives in many ways, and they are continuing to do so. According to the International...
-
Contracting parties to the General Agreement on Tariffs and Trade (1947-94) could use quotas to limit imports of agricultural products from other countries. Under the Uruguay Round (1986-94), import...
-
Australia has over 500 national parks in which commercial activities are prohibited and human activity is strictly monitored. In the interests of visitor safety or environmental sustainability,...
-
Kevin is the sole proprietor of Murphs Golf Shop. During the current year, a hurricane hits the beach near Kevins shop. His business building, which has a basis of $60,000, is damaged. In addition,...
-
You have just begun your summer internship at Omni Instruments. The company supplies sterilized surgical instruments for physicians. To expand sales, Omni is considering paying a commission to its...
-
During its first year of operations, Ketter Company had credit sales of $3,000,000, of which $400,000 remained uncollected at year-end. The credit manager estimates that $18,000 of these receivables...
-
The 2009 financial statements of 3M Company report net sales of $23.1 billion. Accounts receivable (net) are $3.2 billion at the beginning of the year and $3.25 billion at the end of the year....
-
Consider these transactions: (a) Galvao Restaurant accepted a Visa card in payment of a $200 lunch bill. The bank charges a 3% fee. What entry should Galvao make? (b) Stone Company sold its accounts...
-
6- The weather is becoming important to you since you would like to go on a picnic today. If it was sunny yesterday, there is a 70% chance it will be sunny today. If it was raining yesterday, there...
-
9. Find the scale factor and the missing measurements in the similar triangles in each part. (The sketches are not drawn to scale.) a. x 60 9 km b. 70 8 km C. A 3 cm P y B b 4.8 km 6 km 70 50 y 9 cm...
-
According to NBA.com, Lebron James' career free-throw percentageis 74%. However, during the 2016-2017 season, James'free-throw percentage was only 67.4%. Some people may usethis to argue that after...

Study smarter with the SolutionInn App


