Verify that a 20% aqueous solution by mass of sucrose (C 12 H 22 O 11 )
Question:
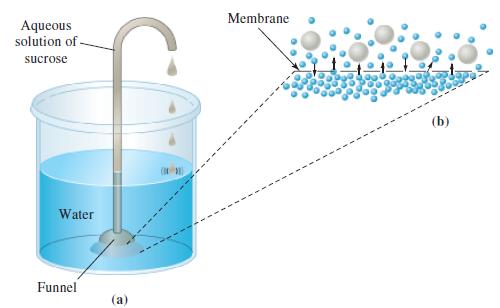
Verify that a 20% aqueous solution by mass of sucrose (C12H22O11) would rise to a height of about 150 m in an apparatus of the type pictured in Figure 14-21.
Figure 14-21
Transcribed Image Text:
Aqueous solution of sucrose Water Funnel (a) Membrane (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Verifying that a 20 aqueous solution by mass of sucrose would rise to a height of about 150 m in an apparatus of the type pictured in Figure 1421 Assumptions The apparatus is semipermeable meaning tha...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A 19.2-g quantity of dry ice (solid carbon dioxide) is allowed to sublime (evaporate) in an apparatus like the one shown in Figure 6.5. Calculate the expansion work done against a constant external...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Shown here is a list published by Electronics Weekly.com of the top five semiconductor companies in the United States by revenue ($ billions). a. Construct a bar chart to display these data. b....
-
Do you think that standard costs are used only in making products like wheel bearings and hamburgers? Think again. Standards influence virtually every aspect of our lives. For example, the next time...
-
Your company is considering two mutually exclusive projects, X and Y, whose costs and cash flows are shown below: The projects are equally risky, and their cost of capital is 12%. You must make a...
-
Thermal regeneration of a fixed-bed adsorber is based on the fact that the adsorption process is exothermic; therefore, heating the saturated adsorbent will result in desorption. The dynamics of the...
-
Hopkins Clothiers is a small company that manufactures tall-mens suits. The company has used a standard cost accounting system. In May 2014, 11,200 suits were produced. The following standard and...
-
Water bottle in a hot car. In the American Southwest, the temperature in a closed car parked in sunlight during the summer can be high enough to burn flesh. Suppose a bottle of water at a...
-
When the stems of cut flowers are held in concentrated NaCl(aq), the flowers wilt. In a similar solution a fresh cucumber shrivels up (becomes pickled). Explain the basis of these phenomena.
-
A 0.72 g sample of polyvinyl chloride (PVC) is dissolved in 250.0 mL of a suitable solvent at 25 C. The solution has an osmotic pressure of 1.67 mmHg. What is the molar mass of the PVC?
-
Suppose Lattin Corp.'s breakeven point is revenues of $1,000,000. Fixed costs are $400,000. Required 1. Compute the contribution margin percentage. 2. Compute the selling price if variable costs are...
-
According to the terms of As will, the city is to become the owner of an apartment building. The net income from the building is to be used to provide bonus awards to public safety employees who are...
-
The Charles Vernon Eames Community Service Center is a nongovernment VHWO financed by contributions from the general public. During 20X5, unrestricted pledges of $900,000 were received, half of which...
-
What are the characteristics of a term bond? a. Term bonds may not exceed 15 years. b. Principal and interest on the entire principal are paid throughout the life of the issue. c. Interest is paid on...
-
Assume for Questions 5 through 9 that the state of Exuberance issued $10,000,000 of 5%, 20-year refunding bonds in 20X5 at par. If the state placed the $10,000,000 in an irrevocable trust as in item...
-
Define the following interfund transaction terms and explain how each is accounted for and reported by a municipality: (a) interfund reimbursement, (b) interfund loan, (c) interfund services provided...
-
A six-column table for JKL Company follows. The first two columns contain the unadjusted trial balance for the company as of July 31, 2013. The last two columns contain the adjusted trial balance as...
-
On average there are four traffic accidents in a city during one hour of rush-hour traffic. Use the Poisson distribution to calculate the probability that in one such hour there arc (a) No accidents...
-
Describe some of the ways that the choice of accounting technique can temporarily depress or inflate earnings.
-
How would rapid inflation affect the accuracy and relevance of a manufacturing companys balance sheet and income statement? Does your answer depend on how much debt the company has issued?
-
In 1970 United Airlines bought four new jumbos for $21.8 million each. These planes were written down straight-line over 16 years to a residual value of $0.2 million each. However, they could have...
-
Mammy is out of the kitchen and it's a great opportunity to get you hands on that cake inside the fridge. Alas, Daddy has the same idea and also rushes to the same end! Your Dad wishes to negotiate...
-
Suppose there are two consumers, A and B. There are two goods, Good X and Good Y. X is the quantity of Good X, and Y is the quantity of Good Y. Suppose the consumers' utility functions are given by:...
-
Julio buys a koi fishpond (and fish to put in it) for his wife on their anniversary. He pays $8000 for the pond and fish with $2000 down. The dealer charges add-on interest of 3.5% per year, and...

Study smarter with the SolutionInn App


