What concentration of formate ion, [HCOO - ] should be present in 0.366 M HCOOH to produce
Question:
What concentration of formate ion, [HCOO-] should be present in 0.366 M HCOOH to produce a buffer solution with pH = 4.06?
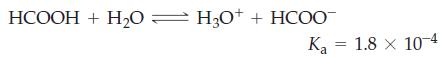
Transcribed Image Text:
HCOOH + H2O=H3O* + HCOO- Ka 1.8 x 10-4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To solve this problem we can use the HendersonHasselbalch equation for ...View the full answer

Answered By

Vineet Kumar Yadav
I am a biotech engineer and cleared jee exam 2 times and also i am a math tutor. topper comunity , chegg India, vedantu doubt expert( solving doubt for iit jee student on the online doubt solving app in live chat with student)
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
What concentration of ammonia, [NH 3 ], should be present in a solution with [NH 4 + ] = 0.732 M to produce a buffer solution with pH = 9.12? For NH 3 , K b = 1.8 x 10 -5 .
-
You work in a research lab with a chemist who asks you to make 500.0 mL of a formic acid/sodium formate buffer solution with pH = 4.10. Formic acid is HCOOH(aq); sodium formate is NaHCOO(s) and is...
-
What mass of sodium formate must be added to 500.0 mL of 1.00 M formic acid to produce a buffer solution that has a pH of 3.50?
-
An investor bought a 70-strike European put option on an index with six months to expiration.The premium for this option was 1. The investor also wrote an 80-strike European put optionon the same...
-
In what financial activities does a corporate treasurer engage?
-
What characteristics of a plant asset make it different from other assets?
-
The transactions for Learning Fun Daycare, Inc., for the month of May 2010 are posted in the following T-accounts. Requirements 1. Calculate account balances. 2. Prepare the trial balance for...
-
A firm reported $250 million in total assets and $140 in debt. It had no interest-bearing securities among its assets. In the income statement it reported $560 million in sales. The firms 80 million...
-
Problem 10: Provide your thoughts and examples on the following. As a guide a couple of paragraphs or about one side on one-page length for each one. a. Why is liquidity risk important? What factors...
-
(A) A 1.00 L volume of buffer is made with concentrations of 0.350 M NaHCOO (sodium formate) and 0.550 M HCOOH (formic acid). (a) What is the initial pH? (b) What is the pH after the addition of...
-
Calculate [OH - ] in a solution that is (a) 0.0062 M Ba(OH) 2 and 0.0105 M BaCl 2 ; (b) 0.315 M (NH 4 ) 2 SO 4 and 0.486 M NH 3 ; (c) 0.196 M NaOH and 0.264 M NH 4 Cl.
-
Find f if f"(x) = 12x 2 + 6x 4, f(0) = 4, and f(1) = 1.
-
The following transactions were completed by the company: a. The company completed consulting work for a client and immediately collected $7,300 cash. b. The company completed commission work for a...
-
Marketing and decision-making go hand in hand. Marketing campaigns are, in fact, created based on knowledge related to how humans behave, what they react to, what makes them happy, etc. Choosing the...
-
The use of pay grades and pay ranges and their relationship to internal alignment and external competitiveness. Why are pay grades and pay ranges necessary?
-
Write Java program to find the factorial of number. Input number should be given by user. ) Write Java program to read a file content line by line. Simple natural language words are very basic index...
-
Save Planning Partners Investors is opening an office in Atlanta, Georgia. Fixed monthly expenses are office rent ($2,000), depreciation on office furniture ($270), utilities ($280), special...
-
Lindsey Smith, Inc., has the following cost structure for the upcoming year: Sales (20,000 units @ $25) ........ $ 500,000 Manufacturing costs: Variable ................ $ 10 per unit Fixed...
-
Planning: Creating an Audience Profile; Collaboration: Team Projects. Compare the Facebook pages of three companies in the same industry. Analyze the content on all available tabs. What can you...
-
The Home Depot reported the following data (in millions) in its financial statements: a. Determine the ratio of net sales to average total assets for The Home Depot for 2007 and 2006. Round to two...
-
Kroger, a national supermarket chain, reported the following data (in millions) in its financial statements for 2007: Total revenue $66,111 Total assets at end of year 21,215 Total assets at...
-
The following selected accounts and their current balances appear in the ledger of Case-It Co. for the fiscal year ended November 30, 2010: 1. Prepare a multiple-step income statement.2. Prepare a...
-
Explain what is meant by the following statement. Please include proper citations in your discussion post The performance report does not compare actual performance to the flexible budget. Therefore,...
-
The informative paragraph must be communicated in a non-biased tone and explain, teach, or inform. The descriptive paragraph must use sensory details to describe a person, place or object. For the...
-
2. Design a two-stage amplifier that produces the following output: V = 3 + 22 + 4V3 a. Draw the block diagram for this system b. Implement the block diagram for this circuit using op-amps. Ensure...

Study smarter with the SolutionInn App


