What is the (a) Degree of ionization and (b) Percent ionization of propionic acid in a solution
Question:
What is the
(a) Degree of ionization and
(b) Percent ionization of propionic acid in a solution that is 0.45 M CH3CH2CO2H?
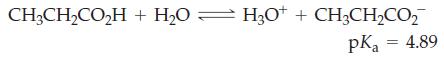
Transcribed Image Text:
CH3CH₂CO₂H + H₂O — H3O+ + CH3CH₂CO₂ pka = 4.89
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a Degree of ionization The degree of ionization is the fraction of the ini...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Exploring the degree of ionization equation in Study Question 127: Calculate the degree of ionization, , for formic acid at the following concentrations: 0.0100 M, 0.0200 M, 0.0400 M, 0.100 M, 0.200...
-
A handbook lists the following formula for the percent ionization of a weak acid. (a) Derive this equation. What assumptions must you make in this derivation? (b) Use the equation to determine the...
-
A 0.400-g sample of propionic acid was dissolved in water to give 50.0 mL of solution. This solution was titrated with 0.150 M NaOH. What was the pH of the solution when the equivalence point was...
-
Gray Corporation began operations on January 1, 2017. The following information is available for Gray Corporation on December 31, 2017. Prepare an income statement, a retained earnings statement, and...
-
What are stock purchase warrants? What are the similarities and key differences between the effects of warrants and those of convertibles on the firms capital structure and its ability to raise new...
-
Born between 1980 and 1990, Generation Next was the topic of Exercise 8.64. In a survey of 500 female and 500 male students in Generation Next, 345 of the females and 365 of the males reported that...
-
You are auditing the financial statements of Aardvark Wholesalers, Inc. (AW), a wholesaler with operations in 12 western states and total revenues of about $125 mil lion. You are carrying out an...
-
Apricot Corporation distributes property ($125,000 basis and $150,000 fair market value) to its sole shareholder, Ellie. The property is subject to a liability of $200,000, which Ellie assumes....
-
What IKEA do We want? Give an executive summary of the case study?
-
What is the (a) Degree of ionization and (b) Percent ionization of ethylamine, C 2 H 5 NH 2 , in a 0.85 M aqueous solution?
-
In the diagram below, the sketch on the far left represents the [OH - ] present in an ammonia solution of molarity c. If the solution is diluted to half its original molarity, which of the sketches...
-
Refer to the Brain and Behavior Evolution (Apr. 2000) study of the feeding behavior of black-bream fish, presented in Exercise 2.162 (p. 96). Recall that the zoologists recorded the number of...
-
Some EU politicians have accused the UK of wanting its cake and eating it. In the context of the referendum vote to leave the EU and the subsequent negotiations on withdrawal, how far would you agree...
-
Classify each of the following histograms as skewed to the left, skewed to the right, or approximately symmetric. 0.30 a. 5000 b. 250 c. 0.25 200 4000 0.20 3000 2000 0.15 0.10 Frequency 150 100 1000...
-
Suppose a bond has a modified duration of 4. By approximately how much will the bonds value change if interest rates a. increase by 50 basis points b. decrease by 150 basis points c. increase by 10...
-
Italian politicians face difficult economic choices. Unemployment is high and growth is sluggish. There is a divide between the more prosperous north and poorer south. As a member of the EU, the...
-
Consider a callable corporate bond with an 8% coupon currently trading at 102. You are asked by management to measure the interest rate risk exposure. How would you proceed?
-
The Chapter Problem for Chapter 3 includes the sample mean of the numbers of chocolate chips in 40 Chips Ahoy reduced fat cookies. The sample mean is f = 19.6 chocolate chips and the sample standard...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
A company reports the following: Net income $120,000 Preferred dividends 20,000 Average stockholders equity 600,000 Average common stockholders equity 500,000 Determine (a) The rate earned on...
-
A company reports the following: Net income $ 180,000 Preferred dividends 12,000 Average stockholders equity 1,200,000 Average common stockholders equity 800,000 Determine (a) The rate earned on...
-
A company reports the following: Net income $340,000 Preferred dividends $40,000 Share of common stock outstanding 40,000 Market price per share of common stock $60.00 (a) Determine the companys...
-
What i. How can a financial institution address the challenges of integrating sustainability into its investment decision-making process? a. By ignoring stakeholder input and concerns b. Through...
-
The task is focused on using Tableau. The question is how would you optimize inventory levels the only columns you have are Product name, Purchase price, Sale price, Sold Quantity, Storage Quantity...
-
AS ACHER Kathy Hansen has a revolving credit account. The finance charge is calculated on the previous month's balance, and the annual percentage rate is 21%. Complete the account activity table for...

Study smarter with the SolutionInn App


