Which aqueous solution from the column on the right has the property listed on the left? Explain
Question:
Which aqueous solution from the column on the right has the property listed on the left? Explain your choices.
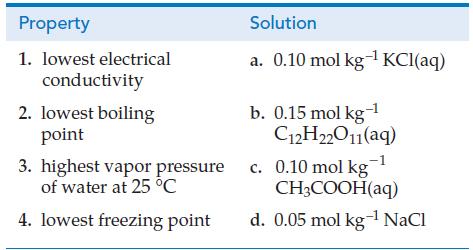
Transcribed Image Text:
Property 1. lowest electrical conductivity 2. lowest boiling point 3. highest vapor pressure of water at 25 °C 4. lowest freezing point Solution a. 0.10 mol kg-¹ KCl(aq) b. 0.15 mol kg-¹ C12H22011(aq) c. 0.10 mol kg1 CH3COOH(aq) d. 0.05 mol kg-¹ NaCl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Property Solution Explanation Lowest electrical conductivity 010 mol kg KClaq KCl is a strong electr...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
For each polynomial function, find (a) (-1), (b) (2), and (c) (0). f(x)=x5x4
-
The T-accounts for Equipment and the related Accumulated Depreciation Equipment for Ada Company at the end of 2014 are shown here. In addition, Ada Companys income statement reported a loss on...
-
On January 1, 2012, the Eugene Company ledger shows Equipment $36,000 and Accumulated Depreciation $13,600. The depreciation resulted from using the straight line method with a useful life of 10...
-
How does make determine whether a target needs to be rebuilt?
-
1. Calculate the 2010 and 2009 liquidity ratios identified using the Ratio Analysis table above. Also calculate the change and the percentage change for the ratios and complete the table. 2. Analyze...
-
Jessa owns a house and lot on 9th Avenue. She sells the house to the Hartley family, who wish to have a conveyance from her that says, to Harriet Hartley for life, remainder to her son, Alexander...
-
A solution (d = 1.159 g/mL) is 62.0% glycerol, HOCH 2 CH(OH)CH 2 OH, and 38.0% H 2 O, by mass. Determine (a) The molarity of glycerol with H 2 O as the solvent; (b) The molarity of H 2 O with...
-
NaCl(aq) isotonic with blood is 0.92% NaCl (mass/volume). For this solution, what is (a) [Na + ]; (b) The total molarity of ions; (c) The osmotic pressure at 37 C; (d) The approximate freezing point?...
-
Wedge Corporation issued $1,500,000 of 10% convertible bonds for $1,620,000 on March 1, 2013. The bonds are dated March 1, 2013, pay interest semiannually on August 31 and February 28, and the...
-
Rudra owns Rudras Computer Store. Rudra sees Nan, a customer, pick up software from a shelf and put it in her bag. As Nan is about to leave, Rudra tells her that she cant leave until he checks her...
-
CopyCat quickly discovers what Ralph has done. The company files a suit against him, alleging that he has gained unauthorized access to its online services in violation of a certain federal statute....
-
Generally, a court can exercise jurisdiction over the residents of the state in which the court is located. (True/False)
-
Eve, a fifteen-year-old minor, buys a smartphone app from EZ Spyware. The contract is fully executed. Eve now wants to disaffirm it. In most states, Eve a. must return only the app to EZ. b. must...
-
Online Services Company (OSC) is an internet service provider. Ads Unlimited, Inc., sends spam to OSCs customers, and some of them then cancel OSCs services. Ads Unlimited is most likely liable for...
-
Why does the marginal revenue product differ between workers in different jobs?
-
Find the intercepts and then graph the line. (a) 2x - 3y = 6 (b) 10 - 5x = 2y
-
Look one more time at Table 4.1, which applies the DCF stock valuation formula to Fledgling Electronics. The CEO, having just learned that stock value is the present value of future dividends,...
-
Look again at Tables 4.3 (Growth-Tech) and 4.7 (Concatenator Manufacturing). Note the discontinuous increases in dividends and free cash flow when asset growth slows down. Now look at your answer to...
-
The constant-growth DCF formula is sometimes written as where BVPS is book equity value per share, b is the plowback ratio, and ROE is the ratio of earnings per share to BVPS. Use this equation to...
-
(10 points) Consider the following code segment. Calculate the time (in sec) required to execute the code segment in (i) a microprocessor runs at 10 MHz and (ii) a microprocessor runs at 12 MHz. [(a)...
-
Required information [The following information applies to the questions displayed below.] Marco Company shows the following costs for three jobs worked on in April. Balances on March 31 Direct...
-
*****Consider a horizontal uniform load of 30 kN/m (with a direction from left to right) along the entire vertical element instead of the load shown***** Determine the pin reactions at pins A,B and C...

Study smarter with the SolutionInn App


