Which of the following names is most appropriate for the molecule with the structure shown below? (a)
Question:
Which of the following names is most appropriate for the molecule with the structure shown below?
(a) Butyl alcohol;
(b) Butan-2 ol;
(c) Butan-1-ol;
(d) Isopentyl alcohol.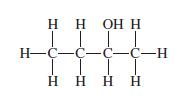
Transcribed Image Text:
НННН ТТТТ H-C-с-с-с-н | | Н НО Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
The correct answer is B Butan2ol accurate...View the full answer

Answered By

Jinah Patricia Padilla
Had an experience as an external auditor in Ernst & Young Philippines and currently a Corporate Accountant in a consultancy company providing manpower to a 5-star hotel in Makati, Philippines, Makati Diamond Residences
5.00+
120+ Reviews
150+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Which of the following names is most appropriate for the molecule CH 3 (CH 2 ) 2 COOH? (a) Dimethyleneacetic acid; (b) Propanoic acid; (c) Butanoic acid; (d) Oxobutylalcohol.
-
Which of the following pairs of compounds has a. The higher boiling point: 1-bromopentane or 1-bromohexane? b. The higher boiling point: pentyl chloride or isopentyl chloride? c. The greater...
-
1. Hannah is applying for a life policy on her girlfriend Sarahs life. The policy is $500,000 and carries a large premium. Hannah is the main earner, so she is concerned about not being able to pay...
-
What is a reporting entity?
-
The following data relate to a $2,000,000, 8% bond issue for a selected semiannual interest period: Bond carrying amount at beginning of period ...... $2,125,000 Interest paid during period...
-
Iris reviewed the scant stack of applications for the newly created security manager position and frowned. There should have been many more than just three applicants for the position. After the...
-
An automobile engine will work best when the back pressure at the interface of the exhaust manifold and the engine block is minimized. Show how reduction of losses in the exhaust manifold, piping,...
-
Mead Bays computer system generated the following trial balance on December 31, 2009. The companys manager knows something is wrong with the trial balance because it does not show any balance for...
-
Describe the value of activity-based customer costing. Explain how ABC can help a firm identify its true low-cost suppliers. Explain driver analysis and its role in process-value analysis. Explain...
-
An 8.129 g sample of MgSO 4 x H 2 O is heated until all the water of hydration is driven off. The resulting anhydrous compound, MgSO 4 weighs 3.967 g. What is the formula of the hydrate?
-
A certain hydrate is found to have the composition 20.3% Cu, 8.95% Si, 36.3% F, and 34.5% H 2 O by mass. What is the empirical formula of this hydrate?
-
What are the two lines that your program must have in order to have logging.debug() send a logging message to a file named programLog.txt?
-
This is a use case for an UBER like application called 'Mishwar' where a UML design class diagram DCD is required for this use case.Cheshire Cat then made additional payments as follows, based upon...
-
Consider the DE = 2x(2000-x). This is a logistic DE. (a) If a(t) is a solution to this DE then what are the possible values for limitt-x(t). (b) If a solution r(t) to this DE also satisfies 0 < x(0)...
-
Your client holds 3 securities in her portfolio: Security Market Value Expected Return Beta MG Corp $10,000 20% 1.35 TC Corp $3,000 -10% 1 PH Corp $8,000 12% 8 What is the Beta of the overall...
-
Muhammad holds 3 securities in his portfolio: Market Value Expected Return Security MG Corp $10,000 TC Corp $3,000 PH Corp $8,000 20% -10% 12% Beta 1.3 18 .8 What is the expected return on the...
-
Ivana has just moved to Edmonton to take up a position in the provincial government, earning $72,000 a year. Edmonton is a big city, so she has decided to lease a car, as well as buy a downtown...
-
Access the miles per gallon data (MPG) on the text website. a. State the null and the alternative hypotheses in order to test whether the average MPG differs from 95. b. Use Excel to calculate the...
-
You have accepted the engagement of auditing the financial statements of the C. Reis Company, a small manufacturing firm that has been your auditee for several years. Because you were busy writing...
-
Grunewald Industries sells on terms of 2/10, net 40. Gross sales last year were $4,562,500, and accounts receivable averaged $437,500. Half of Grunewalds customers paid on the 10th day and took...
-
The D.J. Masson Corporation needs to raise $500,000 for 1 year to supply working capital to a new store. Masson buys from its suppliers on terms of 3/10, net 90, and it currently pays on the 10th day...
-
The Zocco Corporation has an inventory conversion period of 75 days, a receivables collection period of 38 days, and a payables deferral period of 30 days. a. What is the length of the firms cash...
-
The following trial balances are before and after adjustment for Ivanhoe Company at the end of its fiscal year.Prepare the adjusting entries that were made. ( List all debit entries before credit...
-
E3-20 (Algo) Analyzing, Recording, and Evaluating the Effects of Income Statement Transactions [LO 3-3, LO 3-4, LO 3-5] [The following information applies to the questions displayed below.] MyBnB...
-
Required information [The following information applies to the questions displayed below.] The following data reports on the July production activities of the Molding department at Ash Company....

Study smarter with the SolutionInn App


