Which of the following represents MgCl 2 in solution? (a) (b) (c) (d) Legend: OH Cl. Mg.
Question:
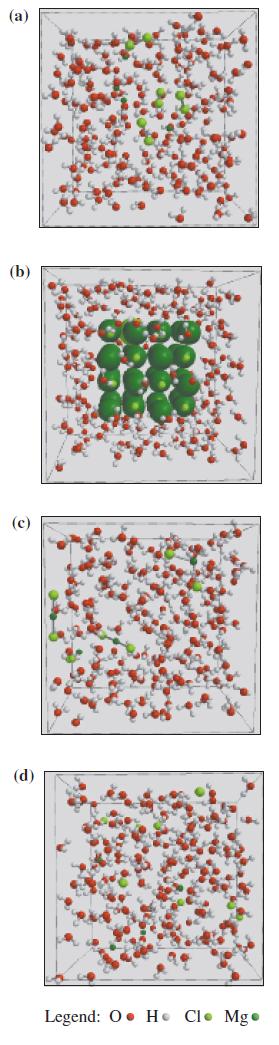
Which of the following represents MgCl2 in solution?
Transcribed Image Text:
(a) (b) (c) (d) Legend: OH Cl. Mg.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
The molecule in the middle represents MgCl2 in solution It is a simple ionic compound so it dissocia...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Which of the following diagrams best represents an aqueous solution of NaF? The water molecules are not shown for clarity. Will this solution be acidic, neutral, or basic?
-
Which of the following pictures best represents a solution made by adding 10 g of silver chloride, AgCl, to a liter of water? In these pictures, the gray spheres represent Ag+ ions and the green...
-
Which of the following pictures best represents an unsaturated solution of sodium chloride, NaCl? In these pictures, the dark gray spheres represent Na+ ions and the green spheres represent chloride...
-
For each polynomial function, find (a) (-1), (b) (2), and (c) (0). f(x) = 2x - 4x + 1
-
Wruck Corporation reported cash provided by operating activities of $450,000, cash used by investing activities of $150,000, and cash provided by financing activities of $80,000. In addition, cash...
-
Distinguish between ordinary repairs and capital expenditures during an assets useful life.
-
A piece of cloth is discovered in a burial pit in the southwestern United States. A tiny sample of the cloth is burned to CO 2 , and the 14 C/ 12 C ratio is 0.250 times the ratio in todays...
-
Nonmonetary Exchanges Holyfield Corporation wishes to exchange a machine used in its operations. Holyfield has received the following offers from other companies in the industry. 1. Dorsett Company...
-
A contractor has to move 15 300 Bank m 3 of wet sandy material in rear dump trucks, which will be loaded by an excavator. Average face depth will be 2.4 m with 60-90 degree average swing angle. Ten...
-
Which of the following ions has the greater charge density? (a) Na + ; (b) F - ; (c) K + ; (d) Cl - .
-
A solution (d = 1.159 g/mL) is 62.0% glycerol, HOCH 2 CH(OH)CH 2 OH, and 38.0% H 2 O, by mass. Determine (a) The molarity of glycerol with H 2 O as the solvent; (b) The molarity of H 2 O with...
-
How might a GC be sure they will receive subcontractor bids on bid day?
-
Gus sends a letter to Jos in which he falsely accuses Jos of embezzling. Joss secretary, Tina, reads the letter. If Jos sues Gus for defamation, Jos will a. win, because Tinas reading of the letter...
-
What determines whether a contract is classified as unilateral or bilateral?
-
Jeff, a fifteen-year-old minor, contracts with Online, Inc., for internet access services. Considering that Jeff is a minor, which of the following is true? a. Online can disaffirm the contract. b....
-
In an exculpatory clause, which of the following statements is true? a. One party agrees that the other party is not mentally incompetent. b. One party releases the other party from liability in the...
-
Some states statutes restrict minors from avoiding certain contracts, including for necessaries. (True/False)
-
Given the analysis of bilateral monopoly, if the passingof minimum wage legislation forces employers to payhigher wage rates to low-paid employees will thisnecessarily cause a reduction in employment?
-
1. As a general strategy, would you recommend that Carl take an aggressive approach to capacity expansion or more of a wait-and-see approach? 2. Should Carl go with the option for one facility that...
-
Alpha Corps earnings and dividends are growing at 15 percent per year. Beta Corps earnings and dividends are growing at 8 percent per year. The companies assets, earnings, and dividends per share are...
-
Look again at the financial forecasts for Growth-Tech given in Table 4.3. This time assume you know that the opportunity cost of capital is r = .12 (discard the .099 figure calculated in the text)....
-
Compost Science, Inc. (CSI), is in the business of converting Bostons sewage sludge into fertilizer. The business is not in itself very profitable. However, to induce CSI to remain in business, the...
-
Required: a-1. Assuming the company can satisfy the annual demand, calculate the contribution margin for each type of sleeve using the table below. a-2. How much operating profit could Williams earn...
-
Input: data: array with n integers Input: n: size of data Output: permutation of data such that data[1] data[2] ... < data[n] 1 Algorithm: StoogeSort 2 if n1 then 3 return data 4 else if n 2 then if...
-
Consider the following four 16b words: 0x8285 0x7644 0x698b Ox8bf What is the checksum that results from adding the four words together? Remember to complement the final result. Blackboard expects a...

Study smarter with the SolutionInn App


