Write a balanced chemical equation for the reaction depicted below. +
Question:
Write a balanced chemical equation for the reaction depicted below.
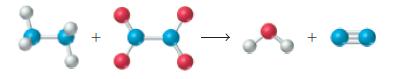
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The balanced chemical equation for the reaction depicted in the image is 2 SiCl4 2 NH4Cl 2 SiF4 2 N...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Write a balanced chemical equation for the reaction that occurs when (a) Calcium metal undergoes a combination reaction with O2(g) (b) Copper(II) hydroxide decomposes into copper(II) oxide and water...
-
Write a balanced chemical equation for the reaction that occurs when (a) Mg(s) reacts with Cl2(g) (b) Barium carbonate decomposes into barium oxide and carbon dioxide gas when heated (c) The...
-
Write a balanced chemical equation for the pentose phosphate pathway in the first two modes depicted in Figure 12.36, where (a) ribose-5-phosphate synthesis is maximized (b) NADPH production is...
-
A company is deciding whether to produce a new gadget at a plant located in a country close to consumers at a higher labor cost and shorter lead time or to outsource it to a country with a low labor...
-
Using the data for Geo-Metrics Corporation in Exercise 14-20, assume that as of December 31, 2008, the M-Labs Inc. stock had a market value of $25 per share and the Spectrum Corp. stock had a market...
-
Refer to Problem 3.1. Data From Problem 3.1 Consider the National Football League data in Table B.1. a. Find a $95 % \mathrm{CI}$ on $\beta_{7}$. b. Find a $95 %$ CI on the mean number of games won...
-
What kinds of leveling mechanisms are present in currentday capitalist society? Are they effective in reducing economic inequality?
-
The Green Division of Frizell Company reported the following data for the current year. Sales ...............$3,000,000 Variable costs ............. 1,950,000 Controllable fixed costs .............
-
McKay operated a large farm on which he grew a variety of vegetables for commercial canners. He also grew a smaller quantity for sale to local retailers and wholesalers as fresh produce. On August...
-
Phosphorus trichloride, PCl 3 is a commercially important compound used in the manufacture of pesticides, gasoline additives, and a number of other products. A ball-and-stick model of PCl 3 is shown...
-
Write balanced equations to represent: (a) The reaction of sulfur dioxide gas with oxygen gas to produce sulfur trioxide gas (one of the reactions involved in the industrial preparation of sulfuric...
-
What are the steps involved in using simulation to evaluate a strategy? Explain each using a specific example of your own or use the DCF illustration in Section 6.4 to identify the steps and explain...
-
What types of government policies can increase long-run living standards?
-
Humidity is a measure of the amount of water vapor in the atmosphere. Why is humidity always very low inside your kitchen freezer?
-
Explain what is meant by a steady state. In the Solow model, which variables are constant in a steady state?
-
Why doesnt the temperature of melting ice rise as the ice is heated?
-
In what sense can you truthfully say that you are a part of every person around you?
-
Suppose an American call option is in the money, so S > X. Demonstrate that the market price of this call (C) cannot be less than the difference between the stock price and the exercise price. That...
-
QUESTION 9 HC-O-C-R R-C-O-CH HC-O-P-O-CH-CH-NH3* O || O a. Phosphatidic acid, Serine O b. Lysophosphatidic acid, Serine, Free FA O c. Lysophosphatidylserine, Free FA O d. 2 Free FAs, Serine, Glycerol...
-
What is free cash flow? Why is it the most important measure of cash flow?
-
If you were starting a business, what tax considerations might cause you to prefer to set it up as a proprietorship or a partnership rather than as a corporation?
-
An investor recently purchased a corporate bond which yields 9 percent. The investor is in the 36 percent combined federal and state tax bracket. What is the bonds after-tax yield?
-
A bungee jumper of mass =85.8 kg steps off a platform a height =50.0 m above the ground. The bungee jumper is connected to the platform by a bungee cord with unstretched length =10.0 m and spring...
-
Calculate the final speed of a spherical ball that rolls without slipping down a 2.00 [m] high incline. The ball starts from rest, has a mass of 0.750 [kg[, and a radius of 4.00 [cm]. Calculate the...
-
An automobile cooling system holds 16 L of water. How much heat does it absorb if its temperature rises from 20 C to 80 C? A 5.0 kg lead ball is dropped into a 2.0 L insulated pail of water initially...

Study smarter with the SolutionInn App


