Write equilibrium constant expressions, K c , for the reactions 2 NO2(g) Zn+ Zn+ (aq) + 2
Question:
Write equilibrium constant expressions, Kc, for the reactions
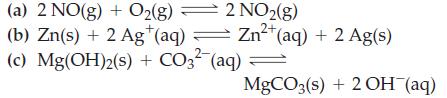
Transcribed Image Text:
2 NO2(g) Zn²+ Zn²+ (aq) + 2 Ag(s) 2+ (a) 2 NO(g) + O2(g) (b) Zn(s) + 2 Ag+ (aq) = (c) Mg(OH)2(s) + CO3²- (aq) = MgCO3(s) + 2OH(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To write the equilibrium constant expressions Kc for the given reaction...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
338+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The solution containing no added KNO3 for Figure 7-1 contains 5.0 mM Fe(NO3)3, 5.0 M NaSCN, and 15 mM HNO3. We will use Davies activity coefficients to find the concentrations of all species in the...
-
Write the expressions for Kc for the following reactions. In each case indicate whether the reaction is homogeneous or heterogeneous.
-
For the synthesis of ammonia the equilibrium constant Kc at 375°C is 1.2. Starting with [H2]0 = 0.76 M, [N2]0 = 0.60 M, and [NH3]0 = 0.48 M, which gases will have increased in concentration and...
-
Both of the following questions are essentially the same. Does the difference in wording seem as though it could affect the way that people respond? Are you in favor of the "Defense of Marriage...
-
Define risk in terms of the cash flows from a capital budgeting project. How can determination of the breakeven cash inflow be used to gauge project risk?
-
Susan permitted Kevin to take her very old grandfather clock on the basis of Kevins representations that he was skilled at repairing such clocks and restoring them to their original condition and...
-
Identify the most appropriate approach and then design. Provide a rationale as to why this approach and design would be most appropriate. Develop a hypothetical research scenario that would...
-
LawnCare USA provides lawn care and landscaping services to commercial clients. LawnCare USA uses activity-based costing to bid on jobs and to evaluate their profitability. LawnCare USA reports the...
-
Provide a definition of a database management system (DBMS) and discuss the purpose(s) in business. Introduce the business scenario to analyze and discuss the benefits of implementing a database...
-
Can you conclude whether the numerical value of K for the reaction 2 ICl(g) I 2 (g) + Cl 2 (g) is greater or less than the numerical value of K for the reaction ICl(g) 1/2 I 2 (g) + 1/2 Cl 2 (g)?...
-
(A) Teeth are made principally from the mineral hydroxyapatite, Ca 5 (PO 4 ) 3 OH, which can be dissolved in acidic solution such as that produced by bacteria in the mouth. The reaction that occurs...
-
A 10-m3 -rigid tank contains R134a at 200 kPa and 20C. Heat is added to this tank until the pressure reaches 300 kPa. How much heat has been added? What is the final temperature of the refrigerant?
-
On the first day of the fiscal year, Lisbon Co. issued $1,000,000 of 10-year, 7% bonds for $1,050,000, with interest payable semiannually. The fiscal year of the company is the calendar year. Prepare...
-
Complete the table below assuming the shrinkage= 18%, and determine the cost of any either waste or borrow volume if you know both cost about 16 US$ per yard, then plot the mass diagram on the...
-
Design a communications link that connects from the moon to a low-earth- satellite. The link requires a rate of 50 Mb/s. The carrier frequency is 60 GHz. Assume temperature is maintained at 200 K....
-
Discuss the carnitine shuttle's purpose, location, primary enzymes and end products involved in B- oxidation. What can go wrong if the carnitine shuttle does not function properly?
-
There are two zero-coupon bonds below: Coupon Term to rate maturity 0% 1 year 10% 2 years Bond A B FV $100 $100 Price $95.24 $107.42 Consider a 2-year coupon bond C with FV = $100, coupon rate = 25%,...
-
Doug has been approached by his broker to purchase a bond of $850. He believes the bond should yield 10%. The bond Pays 7% annual coupon rate and has 12 years left until maturity. What should doug's...
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
Two projects have an identical net present value of $9,000. Are both projects equal in desirability?
-
What are the major disadvantages of the use of the net present value method of analyzing capital investment proposals?
-
What are the major disadvantages of the use of the internal rate of return method of analyzing capital investment proposals?
-
What is the role of resource breakdown structures with managing project costs for a HR Manual?
-
Both Thu and Juan own one-half of the stock of Wren, Inc., a C corporation. Each shareholder holds a stock basis of $175,000. Wren holds accumulated E & P of $300,000. Wren's taxable income for the...
-
Consider the following information for Gelato Supremo for 5.000 units: Gelato Supremo Revenue and Spending For the Month Ended June 30th Costs Cost Per liter Variable Element Fixed Elem ent 5000...

Study smarter with the SolutionInn App


