Write equilibrium constant expressions, K p , for the reactions (a) CS(g) + 4H(g) (b) Ag2O(s)
Question:
Write equilibrium constant expressions, Kp, for the reactions
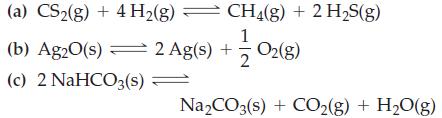
Transcribed Image Text:
(a) CS₂(g) + 4H₂(g) — (b) Ag2O(s) 2 Ag(s) + (c) 2 NaHCO3(s) = CH4(g) + 2 H₂S(g) 12/02 (8) = Na₂CO3(s) + CO₂(g) + H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The equilibrium constant expressions Kp for the reactions in the image are as follows Reaction 1 CS2...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Consider the reactions and their respective equilibrium constants: Use these reactions and their equilibrium constants to predict the equilibrium constant for the following reaction: NO(g) + Br(g) ...
-
The equilibrium constant for the ethane dehydrogenation reaction, is defined as where P(atm) is the total pressure and yi is the mole fraction of the ith substance in an equilibrium mixture. The...
-
The solution containing no added KNO3 for Figure 7-1 contains 5.0 mM Fe(NO3)3, 5.0 M NaSCN, and 15 mM HNO3. We will use Davies activity coefficients to find the concentrations of all species in the...
-
The condensed financial statements of Soule SpA for the years 2016 and 2017 are presented as follows. SOULE SpA Statements of Financial Position December 31 SOULE SpA Income Statements For the Years...
-
How are risk classes often used to apply RADRs?
-
On December 31, 2013, Russell Co. estimated that 2% of its net sales of $360,000 will become uncollectible. The company recorded this amount as an addition to Allowance for Doubtful Accounts. On May...
-
Brad Poison, P.A., collected cash on account from a client for whom the business had provided delivery services one month earlier. Requirements 1. Why did the business fail to record revenue when it...
-
The following information relates to Shirley Corporation's transactions during 2017, its first year of operations. 1. Income before income tax on the income statement for 2017 was $64,000. 2. Income...
-
Given that w = 5 kN/m, F = 17 kN, M = 30 kN.m, a = 3 m, b = 1.5 m and c = 1.5 m, a) Determine the internal normal force (in kN) at point E in the compound beam. b) Determine the internal shear force...
-
For the gas-phase reaction below, the value of K c is 3.4 at 1000 K. What is the value of K p at this temperature? 2 SO2(g) + O2(g) 2 SO3(g)
-
You want to calculate K for the reaction and you have available a K value for the reaction What additional K value do you need, assuming that all K values are at the same temperature? CH4(g) + 2...
-
The rod on the power control mechanism for a business jet is subjected to force F. Determine the moment of this force about the bearing at A. Given: F = 80 N θ 1 = 20 deg a = 150 mm θ 2 =...
-
Given: 1kg = 1000g 1. A pitcher threw a pitch that generated an impulse of 3.25 Ns on the catcher's glove. If the mass of the baseball is 142g, what was the velocity of the pitched baseball when it...
-
1. A car is traveling at 60 mph and needs to come to a stop in 100 feet. What acceleration is required if the brakes are applied after a 0.5-second delay? 2. A ball is rolled around a circular track...
-
Preparing a Job Order Cost Sheet Riverwood Accounting Company has the following account in its cost records: Direct labor Work in Process-Jones Audit 140,000 Services completed 295,771 Project...
-
A bond with a maturity of 14 years sells for $1,088. If the coupon rate is 7.2 percent, what is the yield to maturity of the bond?
-
A 22 kg dolphin moving in the positive direction decelerates from 12.5 to 8.5 m/s in 2.5 s to join another dolphin in play. What average force, in newtons, was exerted to slow him if he was moving...
-
Powers, Inc., has a project that requires an initial investment of $43,000 and has the following expected stream of cash flows: Year 1 ......$20,000 Year 2 ...... 30,000 Required Use Excel to...
-
In July 2013, cnet.com listed the battery life (in hours) and luminous intensity (i. e., screen brightness, in cd/m2) for a sample of tablet computers. We want to know if screen brightness is...
-
Battonkill Company, operating at full capacity, sold 112,800 units at a price of $150 per unit during 2010. Its income statement for 2010 is as follows: The division of costs between fixed and...
-
For the coming year, Tolstoy Company anticipates a unit selling price of $100, a unit variable cost of $30, and fixed costs of $2,100,000. Instructions 1. Compute the anticipated break-even sales...
-
Last year, Douthett Inc. had sales of $2,400,000, based on a unit selling price of $600. The variable cost per unit was $440, and fixed costs were $544,000. The maximum sales within Douthetts...
-
Textra produces parts for a machine manufacturer. Parts go through two departments, Molding and Trimming. The company budgets overhead cost of $256,250 in the Molding department and $216,250 in the...
-
Thalassines Kataskeves, S.A., of Greece makes marine equipment. The company has been experiencing losses on its bilge pump product line for several years. The most recent quarterly contribution...
-
Dorsey Company manufactures three products from a common input in a joint processing operation. Joint processing costs up to the split-off point total $335,000 per quarter. For financial reporting...

Study smarter with the SolutionInn App


