Write Lewis structures for the molecules represented by the following line-angle formulas. (a) C1- (b) HO OH
Question:
Write Lewis structures for the molecules represented by the following line-angle formulas.
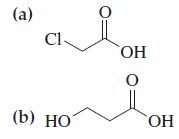
Transcribed Image Text:
(a) C1- (b) HO OH 0 ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Write Lewis structures for the molecules represented by the following line-angle formulas. (a) (b) Cl NH
-
Write Lewis structures for the molecules represented by the following molecular models. (a) (b)
-
Write Lewis structures for the molecules represented by the following molecular models. (a) (b)
-
A firm has initial fund of $800. The market rate of return is 12%. The firm has identified good investment opportunities to invest $640 at an average 25 per cent rate of return. How much dividend...
-
The balance sheets of Tully Corp. showed the following at December 31, 2011, and 2010: Required: a. If there have not been any purchases, sales, or other transactions affecting this machine account...
-
Calculate the available-to-promise row in the followingmatrix. On hand 100 Forecast Customer Orders Master Production Schedule Available-to-Promis Period 1 35 6 50 100 50 100 50 100 50 125 75 175 45...
-
The capital structure of Audiology Associates, Inc., at December 31, 2007 , included 5,000 shares of \(\$ 2\) preferred stock and 100,000 shares of common stock. Common shares outstanding during 2008...
-
A jewelry store makes necklaces and bracelets from gold and platinum. The store has developed the following linear programming model for determining the number of necklaces and bracelets (x1 and x2)...
-
(2 points) The masses mi are located at the points Pi. Answer the following questions. = m1 6, m2 = 12, m3 = 6, and m4 = 12 P(3, 2), P2(6, 2), P3(4, 4), P(4, 3) 1. Find the moment My of the system...
-
Identify the main group that the element X belongs to in each of the following Lewis structures. For the types of molecule shown, give an example that exists. (a) [:XX; :: 2- (c) |:0 x6: (b) :0- ::...
-
Write a plausible Lewis structure for C 3 O 2 , a substance known as carbon suboxide.
-
A candy maker produces mints that have a label weight of 20.4 grams. Assume that the distribution of the weights of these mints is N(21.37, 0.16). (a) Let denote the weight of a single mint selected...
-
Assume that Freedonia, a small country, puts a tariff on steel. How does this affect the market for cars in Freedonia? Be sure to explain your answer.
-
Draw the digraph of the following relations = {(1,2), (2,1), (3,3), (1,1), (2,2)} on X = {1,2,3} b. R = {(1,2), (2,3), (3,4), (4,1)} on X = {1,2,3,4} c. Ron {1,2,3} defined by (x, y) = R, if x y R =...
-
The Industry life cycle has the following phases: introduction, growth, shakeout, maturity and de Identify an industry in Canada that you consider to be in the introduction or growth phase. Tell us...
-
What does Bill Maurer mean by "Money Ecology", and how does this affect the ways people have developed to make payments? Give and discuss examples that contrast different money ecologies and the...
-
Please write a program that calculates the final score of multiple students using different weight of exams and assign their final grade using the following criteria: If the average grade is 90 or...
-
How does application of the predication principle apply to the what question?
-
A stock has had returns of 8 percent, 26 percent, 14 percent, 17 percent, 31 percent, and 1 percent over the last six years. What are the arithmetic and geometric average returns for the stock?
-
This makes no sense at all, said Bill Sharp, president of Essex Company. We sold the same number of units this year as we did last year, yet our profits have more than doubled. Who made the goofthe...
-
These statements cant be right, said Ben Yoder, president of Rayco, Inc. Our sales in the second quarter were up by 25% over the first quarter, yet these income statements show a precipitous drop in...
-
Guochang Li was hired as chief executive officer (CEO) in late November by the board of directors of ContactGlobal, a company that produces an advanced global positioning system (GPS) device. The...
-
1. Label the six phase changes of water on the diagram below (Figure 6.2). Solid Ice Liquid Water Figure 6.2: Phase Changes of Water. (Invisible) Gas Water Vapor
-
6. (4) A roofer pulls a 25.0 kg bundle of shingles up the side of a roof that has a 29.0 pitch. She applies a force to the shingles, counteracting the 18.5 N of friction, to accelerate the shingles...
-
A car, starting from rest, rolls down a 200m high hill, how fast is it going at the bottom of the hill, if the energy transfer has a 70% efficiency? A 1,000,000kg space ship is floating in space,...

Study smarter with the SolutionInn App


