Consider the acids listed and decide on the basis of their structures how you would order them,
Question:
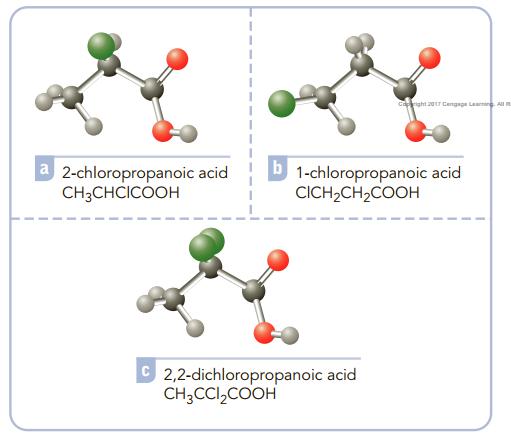
Consider the acids listed and decide on the basis of their structures how you would order them, from weakest to strongest. Explain your reasoning.
Transcribed Image Text:
capright 20r Cengaga Leaning Al 2-chloropropanoic acid CH3CHCICOOH 1-chloropropanoic acid CICH,CH2COOH 2,2-dichloropropanoic acid CH;CCI,COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The electronegative chlorine atom withdraws electron densi...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The directors of Chekani plc, a large listed company, are engaged in a policy of expansion. Accordingly, they have approached the directors of Meela Ltd, an unlisted company of substantial size, in...
-
Shown below are interactions that occur between !tons and polar compounds (cyclopentyl alcohol, dim-ethyl ether, dim-ethyl sulfide . and wafer) Examine me types of inter-molecular forces in which...
-
The following compounds can all react as bases. (a) For each compound, show its conjugate acid. Show any resonance forms if applicable. (b) Rank the conjugate acids in the order you would predict,...
-
In 2006, Walmart was operating more than 6,500 stores with 1.8 million employees around the world. In the early 1990s, Walmart Stores Inc. expanded into Canada, with the purchase of 122 stores from...
-
A manufacturing company produces steel housings for electrical equipment. The main component part of the housing is a steel trough that is made out of a 14-gauge steel coil. It is produced using a...
-
Compute common-size percents for the following comparative income statements (round percents to one decimal). Using the common-size percents, which item is most responsible for the decline in net...
-
A study found that eating a few pieces of chocolate each week can improve your cardiovascular health. Identify the two events described in the study. Do the results indicate that the events are...
-
McGee Company has the following data at December 31, 2010. The available-for-sale securities are held as a long-term investment.Instructions(a) Prepare the adjusting entries to report each class of...
-
Complete a regression analysis to determine total annual fixed and variable costs using the sales revenue and wage costs shown below. Sales Revenue Wage Costs January 24,900 11,300 February 24,200...
-
In an 802.11 network, station A sends one data frame (not fragmented) to station B. What would be the value of the D field (in microseconds) that needs to be set for the NAV period in each of the...
-
Hydrazine, N 2 H 4 (having the structure H 2 NNH 2 ), and its derivatives have been used as rocket fuels. Draw the Lewis electron-dot formula for the hydrazine molecule. Describe the geometries...
-
Saccharin, HC 7 H 4 NO 3 S, is a well-known sweetener. It is a weak acid, and as the sweetener, it is generally supplied as the sodium salt. Here is the structural formula for the saccharin molecule....
-
The longest economic expansion in our history began in _____. a) the spring of 1961 b) the winter of 1982 c) the spring of 1991 d) the fall of 1993
-
1)What is interest rate risk? Which bond has greater interest rate risk, a 10-year zero-coupon Treasury STRIPS or a 10-year Treasury Note? 2)What is the difference between a bond's promised yield and...
-
Instructions: Using the information "CoffeeVille's Risk Areas" from the "Policies & Procedures section in the simulated environment document, create a contingency work plan that you will follow when...
-
You and your partner will imagine a situation in which your business has entered into a contract with a supplier or a customer, and the supplier/customer has breached the contract. Please answer the...
-
Why they are essential for employee motivation, retention, and company sustainability. Write a comprehensive presentation based on the three components listed below. In three parts, outline all the...
-
Instructions: a. Put your answers in a yellow paper. (Use as many pages as needed.) b. Write your name, section and class schedule on each and every page. c. Sign the papers at the bottom, of each...
-
Performance Water Pumps (PWP) is a leading manufacturer of water pumps with high capacity capability. These pumps are used in both civil and private disaster situations. PV/Ws primary market is...
-
Find a polar equation for the curve represented by the given Cartesian equation. 4y 2 = x
-
Consider two hypothetical elements, W and Z. Element W has an electron affinity of 300 kJ/mol, and element Z has an electron affinity of 75 kJ/mol. a. If you have a W ion and a Z ion, from which ion...
-
Suppose that the Pauli exclusion principle were No more than two electrons can have the same four quantum numbers. What would be the electron configurations of the ground states for the first six...
-
Imagine a world in which all quantum numbers, except the l quantum number, are as they are in the real world. In this imaginary world, l begins with l and goes up to n (the value of the principal...
-
You anticipate the receipt of money in 200 days, which you will use to purchase stocks in a particular company. The stock is currently selling for $51 and will pay a $0.5 dividend in 50 days and...
-
1) Based on the stock chart for Michaels Companies Inc, what do you think the short and long-term growth potentials are for this company? (discuss the advantages/disadvantages) Link to the stock...
-
After being drafted in the first round of the NFL draft, a star defensive end invests his signing bonus of $9,827,000.00 in a mutual fund. The fund pays on average 7.00% APR. The player will not...

Study smarter with the SolutionInn App


