Formic acid, HCHO 2 , is used to make methyl formate (a fumigant for dried fruit) and
Question:
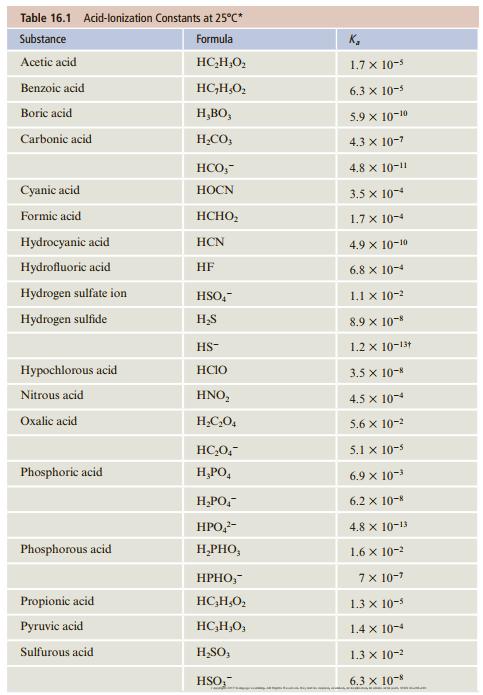
Formic acid, HCHO2, is used to make methyl formate (a fumigant for dried fruit) and ethyl formate (an artificial rum flavor). What is the pH of a 0.12 M solution of formic acid? What is the degree of ionization of HCHO2 in this solution? See Table 16.1 for Ka.

Transcribed Image Text:
Table 16.1 Acid-lonization Constants at 25°C* Substance Formula K, Acetic acid HC,H,O, 1.7 x 10-5 Benzoic acid HC,H,O, 6.3 x 10-5 Boric acid H,BO, 5.9 x 10-10 Carbonic acid H,CO, 4.3 x 10-7 HCO,- 4.8 x 10-11 Cyanic acid HOCN 3.5 x 10-4 Formic acid HCHO, 1.7 x 10-4 Hydrocyanic acid HCN 4.9 x 10-10 Hydrofluoric acid HF 6.8 x 10-4 Hydrogen sulfate ion HSO,- 1.1 x 10-2 Hydrogen sulfide H,S 8.9 x 10- 1.2 x 10-13 HS- Hypochlorous acid HCIO 3.5 x 10-8 Nitrous acid HNO, 4.5 x 10-4 Oxalic acid H,C,O, 5.6 x 10-2 HC,0,- 5.1 x 10-5 Phosphoric acid H,PO, 6.9 x 10-3 H,PO,- 6.2 x 10-8 HPO,- 4.8 x 10-1 Phosphorous acid H,PHO, 1.6 x 10-2 НРНО,- 7x 10-7 Propionic acid HC,H,O, 1.3 x 10-5 Рyruvic acid HC,H,O, 1.4 x 10-4 Sulfurous acid H,SO, 1.3 x 10-2 HSO, 6.3 x 10-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
To solve assemble a table of starting change and equilibrium concentrations ...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Formic acid, HCHO2, is used to make methyl formate (a fumigant for dried fruit) and ethyl formate (an artificial rum flavor). What is the pH of a 0.10 M solution of formic acid? What is the degree of...
-
A 1.24-g sample of benzoic acid was dissolved in water to give 50.0 mL of solution. This solution was titrated with 0.180 M NaOH. What was the pH of the solution when the equivalence point was...
-
A 0.400-g sample of propionic acid was dissolved in water to give 50.0 mL of solution. This solution was titrated with 0.150 M NaOH. What was the pH of the solution when the equivalence point was...
-
If r = (t, t 2 , t 3 ), find r(t) and r(t).
-
The importance of having employees with a healthy work-life balance has been recognized by U.S. companies for decades. Many business schools offer courses that assist MBA students with developing...
-
The Cincinnati Zoo and Botanical Gardens had a record attendance of 1.87 million visitors in 2017 (Cincinnati Business Courier website). Nonprofit organizations such as zoos and museums are becoming...
-
(a) To generate steam at \(60 \mathrm{~atm}\), two processes are proposed: (1) Vaporize water at \(1 \mathrm{~atm}\) and compress the steam at \(60 \mathrm{~atm}\). (2) Pump water to \(60...
-
A man wants to determine whether or not to invest $1000 in a friend's speculative venture. He will do so if he thinks he can get his money back in one year. He believes the probabilities of the...
-
If a sensor (I1:0) is used to count the incoming cars, and another sensor (I1:1) is used to count the outgoing cars, please design a PLC program such that when the number of cars in the parking lot...
-
The Alpine House, Inc. is a large retailer of winter sports equipment. Here is an income statement for the company?s ski department for a recent quarter: THE ALPINE HOUSE, INC.Income Statement?Ski...
-
Boric acid, B(OH) 3 , is used as a mild antiseptic. What is the pH of a 0.015 M aqueous solution of boric acid? What is the degree of ionization of boric acid in this solution? The hydronium ion...
-
C 6 H 4 NH 2 COOH, para-aminobenzoic acid (PABA), is used in some sunscreen agents. Calculate the concentrations of hydronium ion and para-aminobenzoate ion, C 6 H 4 NH 2 COO - , in a 0.080 M...
-
Tai was the sole income beneficiary for life of each of the trusts described below. For each trust, indicate whether and why it was includible in Tais gross estate. a. A trust created under the will...
-
Make an argument that allows you to advocate for a policy change for young teenage boys incarcerated that have no access with education. Title: A good title quickly communicates the contents of the...
-
The soil profile at a site consists of 1.5 m of sand underlain by a 6 m thick clay layer. The water table coincides with the top of the sand layer. The saturated unit weight (Ysat) of the sand and...
-
6. Compute all leading principal minors of the following square symmetric matrices and use them to determine whether the matrix is positive definite, negative definite, or neither. [22-11] a. [4 b. 1...
-
As we have seen in this course, virtually every generation has felt that the existing threat to its privacy was unprecedented in nature. That said, the sheer capacity to collect metadata which exists...
-
Express the equation in exponential form. A) log(16) = 4 That is, write your answer in the form 24 = B. Then indicate what A and B are. B) log, (3125) = 5 That is, write your answer in the form 50 =...
-
If a spaceship is approaching the Earth at 0.100c and a message capsule is sent toward it at 0.100c relative to the Earth, what is the speed of the capsule relative to the ship?
-
Explain briefly what is meant by electronic data interchange (EDI). How does EDI affect a companys audit trail?
-
For the decomposition of one mole of nitrosyl chloride, H = 38 kJ. NOCl(g) NO(g) + 1 / 2 Cl2(g) The activation energy for this reaction is 100 kJ. a. Is this reaction exothermic or endothermic? b....
-
Given the following mechanism for a chemical reaction: H2O2+ I H2O + IO H2O2+ IO H2O + O2 + I a. Write the overall reaction. b. Identify the catalyst and the reaction intermediate. c. With the...
-
Rate constants for reactions often follow the Arrhenius equation. Write this equation and then identify each term in it with the corresponding factor or factors from collision theory. Give a physical...
-
ABC Hospital is one of two hospitals in the community. The Hospital offers all major specialty services (e.g. cardiology, neurology, pulmonology, etc.) The Hospital is approached by a cardiologist...
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
On January 8, the end of the first weekly pay period of the year, Regis Company's employees earned $27,760 of office salaries and $70,840 of sales salaries. Withholdings from the employees' salaries...

Study smarter with the SolutionInn App


