Indicate the molecular geometry around each carbon atom in the compound H;CCH=CHCH,CCH,COOH
Question:
Indicate the molecular geometry around each carbon atom in the compound
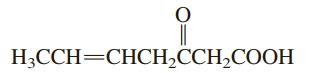
Transcribed Image Text:
H;CCH=CHCH,CCH,COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Numbering the seven carbon atoms from left to right the ...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following samples has a larger mass? O 19.3 g of Au 8.92 10 atoms of Pt Identify the molecular geometry around the three selected atoms. Treat that atoms as the central atom for each...
-
Search Textbo a. chloroform, CHCI3 (carbon is central atom) Lewis structure Total number of electron groups around the central atom Electron geometry Molecular shape b. Carbon tetrachloride, CCI4...
-
The lactic acid molecule, CH3CH (OH) COOH, gives sour milk its unpleasant, sour taste. (a) Draw the Lewis structure for the molecule, assuming that carbon always forms four bonds in its stable...
-
What is the coefficient of sliding friction and what is a representative value for this coefficient for the brittle crust?
-
A coagulation-microfiltration process for removing bacteria from water was investigated in Environmental Science & Engineering (Sept. 1, 2000). Chemical engineers at Seoul National University...
-
On June 15, 2021, Sanderson Construction entered into a long-term construction contract to build a baseball stadium in Washington, D.C., for $220 million. The expected completion date is April 1,...
-
Robert A. Millikan (1865-1953) produced the first accurate measurements on the charge \(e\) of an electron. He devised a method to observe a single drop of water or oil under the influence of both...
-
Mascot Ascots, a retailer of collegiate neckwear, has completed the sales forecast for the coming year. Mascot Ascots maintains an ending inventory level of 65 percent of the following months cost of...
-
You think the political solutions presented by the author are still valid or are other strategies required? "A System at Risk is a comprehensive analysis of the transportation dilemma in the United...
-
What is the Hamming distance for each of the following codewords? a. d (10000, 00000) b. d (10101, 10000) c. d (00000, 11111) d. d (00000, 00000)
-
Draw the cis and trans isomers of the compound 3-methyl-3-hexene.
-
Complete and balance the reaction for each of the following undergoing a combustion reaction. a. 2-methylpentane b. 2,2-dimethylpropane c. 3-methyl-3-hexene d. 3-ethyl-3-propanol e. ethylbenzene
-
What assumptions were made when using Chvorinov's rule to calculate the size of a riser in the manner presented in the text?
-
Through Langham Engineering, Michael Langham marketed his invention, the cross-slope monitor (CSM), as an accessory to John Deere equipment. He had a plan to expand his business to include the CSM as...
-
Pet Food Market to Reach $127.21 Billion By 2027 The global pet food market has four dominant firms that produce more than 50 percent of the market. These firms strive to maintain their dominance by...
-
Unilever was founded in 1929, by the merger of the Dutch margarine producer Margarine Unie and the British soap maker Lever Brothers. The company has grown significantly since then, expanding its...
-
Outline the main stages in the new product development process, identifying the potential sources of failure at each stage.
-
a. What is the quantity of Newfoundland cod fish that fishers catch and the price of Newfoundland cod fish? Is the Newfoundland cod fish stock being used efficiently? Explain why or why not. b. What...
-
Company Z issued bonds with detachable warrants several years ago. Each warrant allows the holder to purchase one share of stock at $30 per share. The stock has a beta of 1.3. a Calculate the...
-
Charles owns an office building and land that are used in his trade or business. The office building and land were acquired in 1978 for $800,000 and $100,000, respectively. During the current year,...
-
a. When 0.10 mol of the ionic solid NaX, where X is an unknown anion, is dissolved in enough water to make 1.0 L of Concept Explorations solution, the pH of the solution is 9.12. When 0.10 mol of the...
-
Which of the following beakers best represents a container of a weak acid, HA, in water? (Water molecules have been omitted for clarity.) ,- = A- = HA A
-
You have 0.10-mol samples of three acids identified simply as HX, HY, and HZ. For each acid, you make up 0.10 M solutions by adding sufficient water to each of the acid samples. When you measure the...
-
Implement the definition of the function rotate ToLeft(), member of the DArray class, so that it rotates all the elements of the array object to the left by one position. Example: [6, 2, 5, 3] [2, 5,...
-
Write the definition of the of the function is Full() as a member function of the DArray class. The function returns true if the array is full, or false otherwise. Functions.cpp 1 #include "DArray.h"...
-
Write a program that will calculate the trajectory of a golf ball thrown with a velocity of 25.6 m/s at an angle of 0 = 35 relative to the horizontal, from a hill that is 2.1 meters above the ground...

Study smarter with the SolutionInn App


