Shown below is a diagram depicting the enthalpy change of a chemical reaction run at constant pressure.
Question:
Shown below is a diagram depicting the enthalpy change of a chemical reaction run at constant pressure.
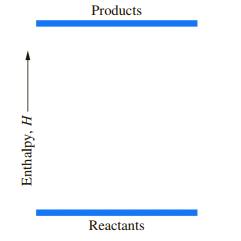
a. Is the reaction exothermic or endothermic?
b. What is the sign of ∆H?
c. What is the sign of q?
d. If the reaction does no work, what is the sign of ∆E for this process?
Transcribed Image Text:
Products Reactants Enthalpy, H-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a Because the enthalpy increases when going from rea...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Thermodynamics and Thermochemistry 105 [Useful information : 1 J= 1kg m's-2, 1 Pa = 1 kg m s; 1 bar 10 Pa] Given that AS (A +C) = 50 eu AS C + D) = 30 eu AS (D - B) = -20 euwhere, eu is entropy unit...
-
14 Thermodynamics and Thermochemistry . The reaction, MgO(s) + C(s) Mg(s) + CO(g ) 18 The entropy change associated with the conversion of 1 kg of ice at 273 K to water vapours at 383 K is for which...
-
1. Which has maximum internal energy at 298 K? a) helium gas c) ozone gas d) equal b) oxygen gas For a gas having molar mass M, specific heat at constant pressure can be given as: YR a) M(Y-1) YRM a...
-
In Exercises find the derivative of the function by the limit process. f(x) = x - 4x + 5
-
The file Energy contains the per capita energy consumption, in kilowatt-hours, for each of the 50 states and the District of Columbia during a recent year. a. Compute the mean, variance, and standard...
-
For each separate case, indicate which type of organization should be formed. a. Sharif, Henry, and Korb want to start a tech firm. They are deciding between an S corporation and a C corporation....
-
A 175-lb woman stands on a vinyl floor wearing stiletto highheel shoes. If the heel has the dimensions shown, determine the average normal stress she exerts on the floor and compare it with the...
-
Assume that you are considering selecting assets from among the following four candidates: Assume that there is no relationship between the amount of rainfall and the condition of the stock market....
-
You are a helping professional working with a 15 year old female name Rosa. Rosa is Hispanic and lives with her mother, father, and two younger brothers. Rosa is bright, high-achieving, and friendly....
-
Can the Count = write(fd, buffer, nbytes); call return any value in count other than nbytes? If so, why?
-
Hydrogen sulfide, H 2 S, is produced during decomposition of organic matter. When 0.5000 mol H 2 S burns to produce SO 2 (g) and H 2 O(l), 281.0 kJ of heat is released. What is this heat in...
-
Consider the following specific heats of metals. Metal .......................Specific Heat copper...................... 0.385 J/(gC) magnesium ...............1.02 J/(gC) mercury...
-
Figure P30.2 shows two electric fields, one in a region of circular cross-section and one in a long flat region. In both cases, the electric field decreases over time. What is the direction of the...
-
Sheridan Inc. presented the following data. Net income $2,672,150 Preferred stock: 55,000 shares outstanding, $100 par, 8% cumulative, not convertible 5,500,000 Common stock: Shares outstanding 1/1...
-
1. Modify the codegiven in the lecture for the up-counter so that it creates an up-down counter with 4 bits. The decision to countup or down is made by the value of the input signal w . If w is 0,...
-
Find the limit. == Let lim f(x) =4 and lim g(x) = -6. Find lim [f(x) - g(x)]. x-9 x-9 x-9
-
30. The stress intensity factor K at a crack is given by K=Cona where o is the far-field stress, a is the crack length, and C is a parameter that depends on the geome- try of the specimen and crack....
-
Logistics Solutions provides order fulfillment services for dot.com merchants. The company maintains warehouses that stock items carried by its dot.com clients. When a client receives an order from a...
-
The revenue equation (in hunderds of millions of dollars) for barley production in a certain country is approximated by R(x)equals=0.0637x^2 + 1.3445x +2.2695 where x is in hundreds of millions of...
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
A vessel containing 39.5 cm3 of helium gas at 25oC and 106 kPa was inverted and placed in cold ethanol. As the gas contracted, ethanol was forced into the vessel to maintain the same pressure of...
-
The volume occupied by a gas depends linearly on degrees Celsius at constant pressure, but it is not directly proportional to degrees Celsius. However, it is directly proportional to kelvins. What is...
-
A sample of 62.3 cm3 of argon gas at 18oC was contained at a pressure of 155 kPa in a J shaped tube with mercury. Later the temperature changed. When the mercury level was adjusted to give the same...
-
AA stock price is $45/share. The stock is expected to pay dividends $2.50 in the coming year. After reviewing all information, you concluded: the price one year from now will be $50/share and you...
-
Fung is planning for the correct after tax and after inflation real rate of return to use. If inflation will be 1.3%, the average rate of return on his investments will be 4.9%, and the tax rate on...
-
In a three - level marketing channel, how many entities ( organization / individual ) are connected?

Study smarter with the SolutionInn App


