Sodium azide, NaN 3 , undergoes the reaction NaN 3 (s) 2Na(s) + 3N 2 (g).
Question:
Sodium azide, NaN3, undergoes the reaction NaN3(s) → 2Na(s) + 3N2(g). Because this reaction is very fast and produces nitrogen gas, NaN3 is used to inflate airplane escape chutes. Sodium azide can be produced through two reaction steps.
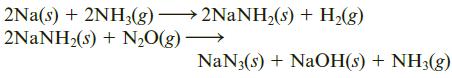
Starting with 1.0 kg of Na, 6.0 kg of NH3, and 1.0 kg of N2O, what is the maximum mass (kg) of sodium azide that can be produced?
Transcribed Image Text:
2Na(s) + 2NH3(g) 2NANH,(s) + H2(g) 2NANH2(s) + N,O(g) - NaN3(s) + NaOH(s) + NH3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
This problem is best approached by first combining the two steps given in the s...View the full answer

Answered By

Jehal Shah
I believe everyone should try to be strong at logic and have good reading habit. Because If you possess these two skills, no matter what difficult situation is, you will definitely find a perfect solution out of it. While logical ability gives you to understand complex problems and concepts quite easily, reading habit gives you an open mind and holistic approach to see much bigger picture.
So guys, I always try to explain any concept keeping these two points in my mind. So that you will never forget any more importantly get bored.
Last but not the least, I am finance enthusiast. Big fan of Warren buffet for long term focus investing approach. On the same side derivatives is the segment I possess expertise.
If you have any finacne related doubt, do reach me out.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following indicate which reaction will occur faster. Explain your reasoning. (a) CH3CH2CH2CH2Br or CH3CH2CH2CH2I with sodium cyanide in dimethyl sulfoxide (b) 1-Chloro-2-methylbutane...
-
In each of the following indicate which reaction will occur faster. Explain your reasoning. (a) CH3CH2CH2CH2Br or CH3CH2CH2CH2I with sodium cyanide in dimethyl sulfoxide (b) 1-Chloro-2-methylbutane...
-
Starting with sodium azide as your source of nitrogen and using any other reagents of your choice, show how you would prepare each of the compounds in Problem 23.18. Compounds in 23.18 (a) (b) (c)...
-
What is the effect of a viscosity (competence) difference between strain markers and the matrix?
-
A bridge with a semielliptical arch spans a river as shown here. What is the clearance 6 ft from the riverbank? 14 ft 50 ft
-
Looking at the TCP header (Figure 24.7), we find that the sequence number is 32 bits long, while the window size is only 16 bits long. Does this mean that TCP is closer to the Go-Back-N or...
-
Exhaust (assumed to have the properties of standard air) leaves the 4-ft-diameter chimney shown in Video V5.4 and Fig. P5.42 with a speed of \(6 \mathrm{ft} / \mathrm{s}\). Because of the wind, after...
-
The summary financial statements of KwikMart Ltd. on December 31, 2014, are as follows: The following errors were made by the inexperienced accountant on December 31, 2013, and were not corrected. l....
-
Problem #3: A steady free jet of water exits a 0.5 inch diameter nozzle with uniform velocity V (point 1). The water jet impinges on a flat plate having a weight of 5 lbf and hinged at the left edge...
-
The cover on a wet-bulb thermometer is soaked in benzene, and the thermometer is exposed to a stream of dry air. The thermometer indicates a temperature of 26oC. Calculate the free stream air...
-
Dinotrogen monoxide, commonly known as laughing gas, can be obtained by cautiously warming ammonium nitrate according to the equation NH 4 NO 3 (s) N 2 O(g) + 2H 2 O(g) If the reaction has a 75%...
-
A sample containing only boron and fluorine was decomposed yielding 4.75 mg of boron and 17.5 mL of fluorine gas (density = 1.43 g/L). What is the empirical formula of the sample compound?
-
(a) Calculate the maximum torque on a 50-turn, 1.50 cm radius circular current loop carrying 50 A in a 0.500-T field. (b) If this coil is to be used in a galvanometer that reads 50 A full scale, what...
-
How does Material Resource Planning (MRP) differ from Enterprise Resource Planning (ERP)?
-
At 3 1 December 2 0 X 9 Folland s receivables totalled 1 2 0 , 0 0 0 . Folland wishes to have an allowance for receivables of 3 , 6 0 0 , which is 2 5 % higher than it was before. During the year...
-
A 1000 kg vehicle climbs a straight 400 m hill at a constant 3 incline. The vehicle accelerates from 3 m/s to 20 m/s. What is the total work done by the vehicle?
-
once a customer has identified a need, two questions arise - what information is required to satisfy the need, and 1. how important is the need? 2. where can i get the information? 3. when does the...
-
A 3 . 0 kg mass hangs at one end of a rope that is attached to a support on a railroad car. When the car accelerates to the right, the rope makes an angle of 1 . 5 degree with the vertical, as shown...
-
On December 1, 2013, Lavender Manufacturing Company (a corporation) purchased another company's assets, including a patent. The patent was used in Lavender's manufacturing operations; $49,500 was...
-
What are the 5 Cs of marketing channel structure?
-
Hydrogen peroxide, H2O2, is a colorless liquid. A concentrated solution of it is used as a source of oxygen for rocket propellant fuels. Dilute aqueous solutions are used as a bleach. Analysis of a...
-
Nitric acid, HNO3, is a colorless, corrosive liquid used in the manufacture of nitrogen fertilizers and explosives. In an experiment to develop new explosives for mining operations, a sample...
-
Hydrogen cyanide, HCN, is a volatile, colorless liquid with the odor of certain fruit pits (such as peach and cherry pits). The compound is highly poisonous. How many molecules are there in 56 mg...
-
Estimate the final temperature in Celsius of the combination if I drop 5 rolls of pennies at 2 0 C ( each roll is 5 0 pennies stacked together; assume they are pure copper ) into a cup of recently -...
-
A 100 gram mass is hung of then end of a cantilever beam . The beam is made of brass, and is 30 cm long (), 2 cm wide, and 3 mm thick. Find the resulting deflection ()
-
The nitrogen-vacancy (NV) and silicon-vacancy (SiV) defects in diamond can lead to many exciting quantum nanotechnologies in physical and biological sciences, including single-photon sensors,...

Study smarter with the SolutionInn App


