(a) By considering Fig. 27.9, suggest a method for the separation of Am from U, Np and...
Question:
(a) By considering Fig. 27.9, suggest a method for the separation of Am from U, Np and Pu.
(b) What would you expect to happen when a solution of NpO2(ClO4)2 in 1Μ HClO4 is shaken with Zn amalgam and the resulting liquid decanted from the amalgam and aerated?
Figure 27.9.
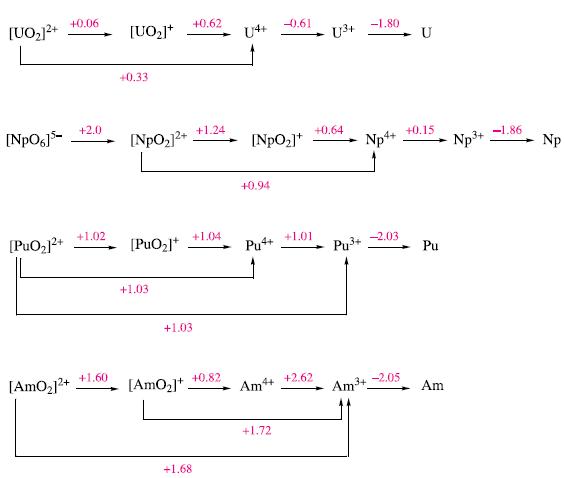
Transcribed Image Text:
[UO₂]²+ [NpO6]5- [PuO₂]²+ [AmO₂]²+ +0.06 +2.0 +1.02 +1.60 [UO₂]+ +0.33 [NpO₂]²+ [PuO₂]* +1.03 +1.03 [AmO₂]+ +0.62 +1.24 +1.04 +1.68 +0.82 [NpO₂]* +0.94 Pu4+ Ĵ Am4+ -0.61 +1.72 +1.01 +2.62 U³+ +0.64 Pu³+ -1.80 Np4+ Am ³+ -2.03 -2.05 U +0.15 Pu Am 3+ Np³+ -1.86 Np
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Method for the separation of Am from U Np and Pu From the given redox potentials standard reduction potentials for various oxidation states of urani...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What would you expect to happen when chlorine gas, Cl2, at 1 atm pressure is bubbled into a solution containing 1.0 M F and 1.0 M Br at 25oC? Write a balanced equation for the reaction that occurs.
-
In Chapter 7, (Data in Tompkins County Bridges 2014) we found a relationship between the age of a bridge in Tompkins County, New York, and its condition as found by inspection. But we considered only...
-
What would you expect to happen to the risk-free rate and equity returns when a segmented country opens its capital markets to foreign investment?
-
There is a crop that the value p next month is random. The value can be either small p=1 or large P=3 depending on the weather. Half of people are optimists believing the value of the crop will be...
-
Describe how the six elements of a standard unit cost increase cost comparability and understandability.
-
Refer to the Lincolnville School District bus data. First, add a variable to change the type of engine (diesel or gasoline) to a qualitative variable. If the engine type is diesel, then set the...
-
An engineer prepares a report to evaluate a project using PW and IRR. Just before submitting the report, he spills coffee on it, making the first digit of the 2-digit IRR unreadable. The second digit...
-
Analyze each transaction listed in the table that follows and place X's in the appropriate columns to indicate the transaction's classification and its effect on cash flows using the indirectmethod....
-
Implement the Boolean function F(A.B.C.D) - (0,1,3,4,8,9,15) with a. a multiplexer b. a decoder Question 2 Draw the NAND gate representation of G A+ B'C' + DC. [10 marks] [5 marks] [5 marks]
-
Vasovist is the tradename of a Gd(III) complex which was the first intravascular contrast agent (see Box 4.3) approved in the EU for use in magnetic resonance angiography. Interactions between the...
-
(a) If, in an electron-transfer process, there is both electron and ligand transfer between reagents, what can you conclude about the mechanism? (b) Explain why very fast electron transfer between...
-
What are the terms of federal funds? Why are these terms often misleading?
-
1 Explain why the larger waves seen on the oscilloscope represent the ventricular contraction.
-
what molecule is shown? what is the primary function of this molecule in a cell? O 01110 N O 111110 O phosphoanhydride bonds CH, O 11P100 0-CH H H OH OH H N C N=C NH N H
-
Calculate the total accumulated value four years from now of $3,000 invested today and $9,000 invested two years later, implied by the table of current spot rates below. (Round your answer to the...
-
What did Itay Talgam mean when he intimated that great leadership can involve "doing without doing"?...
-
Elucidate the intricate mechanisms underlying transcriptional regulation within eukaryotic cells, detailing the orchestrated interplay among transcription factors, enhancers, and chromatin remodeling...
-
Gonzaga University professors conducted a study of television commercials and published their results in the Journal of Sociology, Social Work and Social Welfare (Vol. 2, 2008). The key research...
-
Jax Incorporated reports the following data for its only product. The company had no beginning finished goods inventory and it uses absorption costing. $ 57.30 per unit $ 10.30 per unit $ 7.80 per...
-
Dimethoxyethane (DME) is a polar aprotic solvent often used for S N 2 reactions. Propose a plausible synthesis for DME using acetylene and methyl iodide as your only sources of carbon atoms....
-
Using compounds that possess no more than two carbon atoms, propose a plausible synthesis for the following compound.
-
Assign an IUPAC name for each of the following compounds. a. b. c. d. e. f. g. SH
-
(4.3) Express 600 x 212 e-x12 dx in terms of the gamma function.
-
1. We've multiplied linear functions and divided linear functions by linear functions. What happens when you take the square root of a linear function? Use desmos to graph all of the following on one...
-
Let's say the function f = X(-3,3] L (R) is given. Find real number x and positive number r. Also find the Hardy-Littlewood maximum function and explain why. Also, check that the 3||f1 m({xER:...

Study smarter with the SolutionInn App


