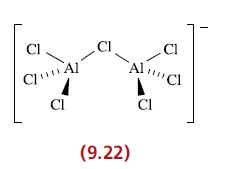
(a) Describe the bonding in the [Al 2 Cl 7 ] anion (9.22). (b) Equilibria 9.82...
Question:
(a) Describe the bonding in the [Al2Cl7]− anion (9.22).
(b) Equilibria 9.82 and 9.83 describe part of the NaCl– Al2Cl6 system; additionally [Al3Cl10]− is present. Write an equation to show how [Al3Cl10]− may be formed, and suggest a structure for this anion.

Transcribed Image Text:
Cl CAI CI CI Cl AlCl A CI (9.22)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a Bonding in the Al2Cl7 anion 922 The Al2Cl7 anion is formed when two aluminum Al atoms bond with se...View the full answer

Answered By

Faith Chebet
In the past two years many of my students have excell with the knowledge I taught them.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Describe the bonding in the CO32- ion using the LE model. How would the molecular orbital model describe the bonding in this species?
-
Describe the bonding in the O3 molecule and the NO2- ion using the LE model. How would the molecular orbital model describe the bonding in these two species?
-
Describe the bonding in O3, using molecular orbital theory. Compare this with the resonance description.
-
The data in CPU reflect the annual values of the consumer price index for all urban consumers (CPI-U) in the United State over the 54-year period 1965 through 2018, using 1982 through 1986 as the...
-
Refer to your solution for the preceding problem regarding McCartney Company. In the preceding problem, McCartney Company manufactures guitars. The company uses a standard, job- order cost accounting...
-
State the five stage of a group's development..
-
Diageo North America, Inc., the owner of the Bulleit brand family of whiskeys, sued competitor W.J. Deutsch & Sons Ltd. for allegedly infringing upon the trade dress of Diageos Bulleit bottles....
-
In the audit of Price Seed Company for the year ended September 30, the auditor set a tolerable misstatement of $50,000 at an ARIA of 10%. A PPS sample of 100 was selected from an accounts receivable...
-
We need to map following EERD into RELATIONAL MODEL. Map the following EERD into relational model ArtistNumber Artist Name N FormationDate Group Group-Member SoloPerformer BirthDate JoinedDate...
-
(a) Give three examples of commonly used ionic liquids. What general properties make ionic liquids attractive in green chemistry? Are the properties of the liquid itself all that determines whether...
-
Using your answers to problem 7.11, write down expressions for the solubility (in mol dm 3 ) of (a) AgCl, (b) CaCO 3 (c) CaF 2 in terms of K sp . Data from Problem 11 Write down expressions for Ksp...
-
On June 15, 2017, Allen sold land held for investment to Stan for $50,000 and an installment note of $250,000 payable in five equal annual installments beginning on June 15, 2017, plus interest at...
-
Macro Corporation and Micro Company combine, and a new organization, MM, Inc., takes their place. This is a. a consolidation. b. a merger. c. a purchase of assets. d. a purchase of stock.
-
Hasty Pudding, a dessert caf, features an area near its dining room where its patrons can leave their coats and other possessions while on the premises. To limit liability for loss or damage to these...
-
First Brownsville Company borrowed funds from Beach Community Bank to build and operate a mini-warehouse storage business. The loan was secured by a mortgage. First Brownsville defaulted on the...
-
McKenna leases ten thousand square feet of space in Midtown Lofts to Home Gallery, an interior design firm, for a five-year term. Less than two years into the term, Home Gallery ceases doing...
-
Douglas and Archondoula Edmonds borrowed funds from Chase Bank USA, National Association (N.A.), secured by a mortgage on real property in Cape Coral, Florida. The mortgage required the lender to...
-
As a result of its export sales to customers in Switzerland, the Lenox Company has had Swiss franc denominated revenues over the past number of years. In order to gain protection from future exchange...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
Identify the products from the reaction between the following pairs of reagents. In each case identify the species which are acting as a Lewis acid or a Lewis base in the reactions. (a) CsF + BrF 3...
-
Use the data in Table 5.5 to calculate the enthalpy change for the reaction of iodine with phenol. Table 5.5. TABLE 5.5 Drago-Wayland parameters for some acids and bases* E Acids Antimony...
-
For each of the following processes, identify the acids and bases involved and characterize the process as complex formation or acidbase displacement. Identify the species that exhibit Brnsted...
-
How do advancements in synthetic biology and genome editing technologies, such as CRISPR-Cas systems and base editing tools, expand the capabilities of recombinant DNA technology to engineer complex...
-
Investors can allocate their wealth among three assets: one risk - free asset and two risky assets ( Stock A and Stock B ) . Both Investors 1 and 2 are rational, mean - variance optimizers and they...
-
QUESTIONS: 1. Write your opinion on what is the main cause of the oil spill. 2. Based on your answer in paragraph 1, explain and justify your opinion (what makes you think that is the main cause). 3....

Study smarter with the SolutionInn App


