(a) Describe the bonding in the M 2 diatomics (M = Li, Na, K, Rb, Cs) in...
Question:
(a) Describe the bonding in the M2 diatomics (M = Li, Na, K, Rb, Cs) in terms of valence bond and molecular orbital theories.
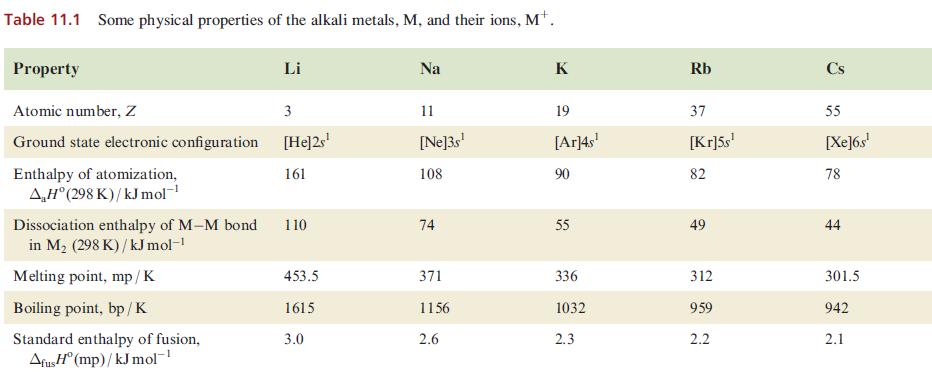
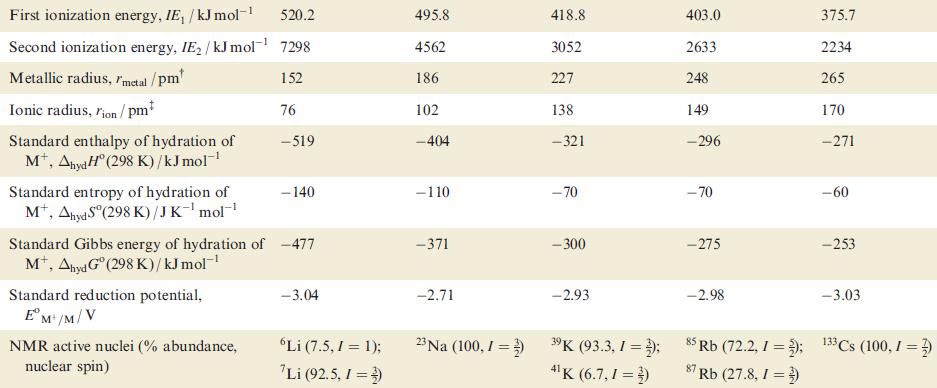
(b) Account for the trend in metal–metal bond dissociation energies given in Table 11.1.
Transcribed Image Text:
Table 11.1 Some physical properties of the alkali metals, M, and their ions, Mt. Property Atomic number, Z Ground state electronic configuration [He]2s¹ 161 Enthalpy of atomization, AH°(298 K)/kJ mol-¹ Dissociation enthalpy of M-M bond in M₂ (298 K)/kJ mol-¹ Melting point, mp/K Li Boiling point, bp/K Standard enthalpy of fusion, Afus H (mp)/kJ mol-¹ 3 110 453.5 1615 3.0 Na 11 [Ne]3s¹ 108 74 371 1156 2.6 K 19 [Ar]4s¹ 90 55 336 1032 2.3 Rb 37 [Kr]5s¹ 82 49 312 959 2.2 Cs 55 [Xe]6s¹ 78 44 301.5 942 2.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a Bonding in M2 diatomics M Li Na K Rb Cs in terms of valence bond and molecular orbital theories Valence Bond Theory In valence bond theory chemical bonds are formed by the overlap of atomic orbitals ...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the Divergence Test to determine whether the following series diverge or state that the test is inconclusive. k k=1 k2 + 1
-
What is the quadratic formula? Start with x=
-
Quantity A Quantity B -(x - 3)/4 y a. Quantity A is greater. b. Quantity B is greater. c. The two quantities are equal. d. The relationship...
-
If we define the median of a sequence as a number so that exactly as many elements come before it in the sequence as come after it, fix the program in 4.6.3 so that it always prints out a median....
-
What is the key feature of activity-based responsibility accounting? Briefly explain.
-
Calculate the one-way ANOVA and determine its statistical significance for the following data: Group 2 4 23 30 7.2 34 29 26 5.8 6.0 6.5 7.0 14 12 15 12 15
-
Which of the following is an activity not usually associated with forensic accounting and fraud examination consulting and litigation support? 1. A. Assessing fraud risk associated with internal...
-
Karel Svoboda, a credit officer for Rogue Bank, evaluated and approved his employers extensions of credit to clients. These responsibilities gave Svoboda access to nonpublic information about the...
-
How would you import a relational model design and generate a logical model from a relational model?
-
Magnesium hydride possesses a rutile lattice. (a) Sketch a unit cell of rutile. (b) What are the coordination numbers and geometries of the Mg and H centres in this structure?
-
Write equations for the following processes, noting appropriate conditions: (a) Electrolysis of water; (b) Electrolysis of molten LiH; (c) CaH 2 reacting with water; (d) Mg treated with dilute nitric...
-
What are the direct and indirect costs of bankruptcy? Briefly explain each.
-
Analytics Corporation acquires substantially all of the assets of Big Data Company by direct purchase. There is no change in either legal entity. This is a. a consolidation. b. a purchase of assets....
-
Stocks represent the purchase of corporate ownership. (True/False)
-
On an assignment of a lease, who is obligated to pay the rent?
-
Employers are required by federal statute to establish health insurance and pension plans. (True/False)
-
A general partnership cannot exist unless a certificate of partnership is filed appropriately in a state. (True/False)
-
White Company was incorporated on January 2, Year 1, and commenced active operations immediately. Common shares were issued on the date of incorporation and no new common shares have been issued...
-
Write a paper about medication error system 2016.
-
Superacids are well established but superbases also exist and are usually based on hydrides of Group 1 and Group 2 elements. Write an account of the chemistry of superbases.
-
Calculate the equilibrium concentration of H 3 O + in a 0.10 M solution of butanoic acid (K a = 1.86 10 5 ). What is the pH of this solution?
-
In their paper The strengths of the hydrohalic acids (J. Chem. Educ., 2001, 78, 116), R. Schmid and A. Miah discuss the validity of literature values of the pK a values for HF, HCl, HBr, and HI. (a)...
-
Porters 5 force ( industry attractiveness) on Carrefour attempting to to enter new foreign market of India,with citation and reference. 12 Cs key factors in relation to the chosen new market, with...
-
At the foreign currency dealer, you are able to see the following direct quote: TZS/US$ 1,500 - 1,600. Required i. Convert the direct quote into indirect quote version. ii. If the dealer buys and...
-
In January the company produced 3,340 units using 2,792 direct labour-hours. During the month, the actual direct labor cost was $63,813. Direct labour 0.8 The labour rate variance for January is?

Study smarter with the SolutionInn App


