(a) Give descriptions of the bonding in ClO 2 and [ClO 2 ] (17.24 and 17.35),...
Question:
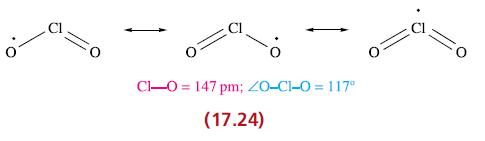
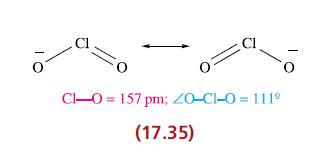
(a) Give descriptions of the bonding in ClO2 and [ClO2]− (17.24 and 17.35), and rationalize the differences in Cl—O bond lengths.
(b) Rationalize why KClO4 and BaSO4 are isomorphous.
Transcribed Image Text:
O Cl-0= 147 pm; 20-C1-0 = 117° (17.24)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a Bonding in ClO2 and ClO2 ClO2 Chlorine dioxide Chlorine dioxide is a paramagnetic bent molecule with a structure like AX2E The central chlorine atom ...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The oxyanion of nitrogen in which it has the highest oxi-dation state is the nitrate ion (NO3-). The corresponding oxyanion of phosphorus is PO43-. The NO43- ion is known but is not very stable. The...
-
The NMR spectra below are for the organic compounds C6H12 and C4H10O. Deduce the structures for these compounds. See Exercise 70 for a discussion of the bonding in organic compounds. The structure of...
-
The melting and boiling points of the titanium tetrahalides are given below. Rationalize these data in terms of the bonding in and the intermolecular forces among these compounds. bp (C) mp (C) 284...
-
This is one question with sub parts please solve this question step by step please also write the little explanation to solve the question Consider the following complex numbers: z, = 3+ 3i z2 = 3 +...
-
In the Home Depot 2009 financial statements in Appendix A at the end of this textbook, read note 1. Find the information about Home Depots international store locations. a. In what countries (other...
-
What is metadata? How does this term pertain to a database?
-
Using only the factor formulas given in Table 2.6, derive Equation 7.6 starting with Equation 7.3. TABLE 2.6 Summary of Discrete Compounding Interest Factors. To Find Given Factor Symbol Name P F...
-
1. Would Lyles claim of racial discrimination be for intentional (disparate-treatment) or unintentional (disparate-impact) discrimination? Explain. 2. Can Lyle establish a prima facie case of racial...
-
For each of the following matrices A Maxn (R), test A for diagonal- izability, and if A is diagonalizable, find an invertible matrix Q and a diagonal matrix D such that Q-1AQ = D. (a) (63) 2 1 3 (b)...
-
Figure 17.15 shows a FrostEbsworth diagram for chlorine. (a) How is this diagram related to the potential diagram for chlorine in Fig. 17.14? (b) Which is the most thermodynamically favoured species...
-
The electronic spectra of mixtures of CH 2 Cl 2 solutions (each 0.993 mmol dm 3 ) of I 2 and the donor D shown in the diagram after the table were recorded for different volume ratios of the two...
-
Design a circuit in which the following series resonant frequencies are switch-selectable: 500 kHz, 1000 kHz, 1500 kHz, 2000 kHz.
-
An asset was bought for OMR . 1 2 , 0 0 0 . Its accumulated depreciation is OMR . 7 , 7 0 0 and the rate of depreciation is 2 0 % . What is the amount of depreciation charge for the current...
-
4. Below are some accounts and their balances for a merchandising business. The ending inventory figure is $15,600. From this data, calculate the cost of goods sold figure.. Accounts Bank Accounts...
-
The assets most commonly used for security by asset-based lending companies are: accounts receivable and inventory inventory and equipment land and buildings equipment and buildings Tim Atte 15 F
-
Teresa drove a company car 1 5 , 0 0 0 personal kilometres in 2 0 2 0 . The total kilometres driven for the year were 2 5 , 0 0 0 kilometres. The actual operating costs for this automobile were $ 8 6...
-
D Question 16 (15 points) Sable systems believes that there is a strong relationship between its monthly factory maintenance costs and the number of hydrogen fuel cells produced in its manufacturing...
-
Consider the sample of 65 customer satisfaction ratings given in Table 12.11. Carry out a chi-square goodness-of-fit test of normality for the population of all customer satisfaction ratings. Recall...
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
Which metal carbonyl in each of (a) [Fe(CO) 4 ] 2 or [Co(CO) 4 ] , (b) [Mn(CO) 5 ] or [Re(CO) 5 ] should be the most basic towards a proton? Explain your answer.
-
What conclusions can you draw from the bonding and reactivity of dihydrogen bound to a low-oxidation-state d-block metal that might be applicable to the bonding of an alkane to a metal? What...
-
Using the 18-electron rule as a guide, indicate the probable number of carbonyl ligands in (a) [W( 6 -C 6 H 6 )(CO) n ], (b) [Rh( 5 -C 5 H 5 )(CO) n ], (c) [Ru 3 (CO) n ].
-
Investigate some applications of exponential and logarithmic functions in science and engineering. Tell us why you think it is interesting. Discuss a topic different from the ones mentioned already...
-
The heat of vaporization AH, of dichloromethane (CH2Cl2) is 28.0 kJ/mol. Calculate the change in entropy AS when 280. g of dichloromethane condenses at 39.8 C. Be sure your answer contains a unit...
-
Evaluate the integral, ez dz - z(z i)(z 3) - where is the circle of radius 2 centered at the origin. Hint: use Cauchy's theorem or Cauchy's integral formula.

Study smarter with the SolutionInn App


