(a) Stability constants for the formation of [M(18-crown-6)] + complexes in acetone are given below. Comment critically...
Question:
(a) Stability constants for the formation of [M(18-crown-6)]+ complexes in acetone are given below. Comment critically on these data.
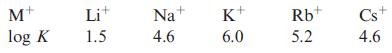
(b) Of the salts NaNO3, RbNO3, Cs2CO3, Na2SO4, Li2CO3, LiCl and LiF, which are soluble in water? Using LiCl and LiF as examples, discuss factors that contribute to the solubility of a salt.
Transcribed Image Text:
M+ log K Lit 1.5 Na+ 4.6 K+ 6.0 Rb+ 5.2 Cs+ 4.6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Stability constants for the formation of M18crown6 complexes in acetone M Li Na K Rb Cs log K 15 46 60 52 46 In these stability constant data a higher log K value indicates a stronger tendency for t...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A The fraud triangle consists of three factors that contribute to management and employee fraud. Explain the relationship among these three factors. (4 Marks) B. Indicate whether the following risks...
-
Using methanol (CH3OH) as a fuel source, write a balanced reaction for the formation of H2. Do the same for C10H22 (a component of diesel fuel). For an H2 fuel cell, why would it be better to use...
-
The equilibrium constant (KP) for the formation of the air pollutant nitric oxide (NO) in an automobile engine at 530°C is 2.9 Ã 10-11: (a) Calculate the partial pressure of NO under these...
-
Describe three ways in which a gradual increase in an extracellular signal can be sharpened by the target cell to produce an abrupt or nearly all-or none response.
-
RELY Cleaning Services, Inc., was started a number of years ago by Rick Ely to provide cleaning services to both large and small businesses in their home city. Over the years, as local businesses...
-
The spectral transmissivity of a glass cover used in a solar collector is given as Solar radiation is incident at a rate of 950 W/m 2 , and the absorber plate, which can be considered to be black, is...
-
Reconsider Problem 65. Plot a graph of capital recovery cost versus useful life for lives 1 to 25 by 1. Data from problem 65 Crush Autosmashers can purchase a new electromagnet for moving cars at a...
-
Implement the following LP problem in a spreadsheet. Use Solver to solve the problem and create a Sensitivity Report. Use this information to answer the following questions: MAX: 4X1 + 2X2 Subject...
-
A cylindrical silo topped by a hemispherical vault of 1920 m3 was built. The production cost per m2 is double in the vault than in the cylindrical part. The cost is $8000 pesos per m2 in the vault If...
-
Write down the formulae of the following ions: (a) Superoxide; (b) Peroxide; (c) Ozonide; (d) Azide; (e) Nitride; (f) Sodide.
-
The first list opposite contains the formula of a group 1 metal or metal compound. Match these to the descriptions given in the second column. List 1 LigN NaOH Cs Cs70 List 2 Reacts explosively with...
-
Describe the types of financial ratios and other financial performance measures that are used during a ventures successful life cycle. Who are the users of financial performance measures?
-
Job stress is a major problem for employees working in many organizations today. Discuss some of the job stressors. What can a manager do to reduce stressors for employees?
-
Mintzberg suggests that managerial roles should encompass interpersonal, decisional, and informational roles. Clearly, this is an idealized vision of the manager. They are encouraged to encompass all...
-
Technological developments deskill the global workforce. For example, factory-built, flat-pack furniture cut out the role of experienced carpenters. Similarly, with some vehicles having on-board...
-
In 2013, a clothing factory in Bangladesh collapsed, killing 1,138 people. Some 27 global brands, including Walmart and Benetton, were using the factory. One year on, these two corporations were...
-
Scott Emmons was working for Neiman Marcus, the luxury retailer, as an enterprise architect when he realized a big gap in how their stores were handling technology. Customers were showing up with...
-
Distinguish between unrestricted and restricted contributions of a charitable organization.
-
What is a lobbyist in US? How did this term emerge?
-
Discuss how hydrogen bonding in inorganic compounds can be characterized using infrared spectroscopy. How does the strength of a hydrogen bond affect (a) OH stretching modes (b) OH bending modes
-
Why are the reported errors on the NH bond lengths determined in (NH 4 ) 2 SeO 4 by single-crystal X-ray diffraction much larger than those on the SeO bond lengths?
-
Use the data in Table 8.3 to estimate the OO stretching wavenumber expected for a compound believed to contain the oxygenyl species O 2 + . Would you expect to observe this stretching vibration in...
-
Problem 3 (10 points) On January 3, 2023, Stem Corporation retired $10,000,000 of bonds by repurchasing them for $10,100,000 (at 101). Assume that the bond interest payment on December 31, 2022 has...
-
The Matilda Corporation purchased Goodwill Company, which belongs to the dairy industry. Given the following information for Goodwill Company, Matilda asked you to compute the proposed purchase...
-
The Corporate Comparison Instructions: 1. 2. Choose two corporations in the same industry (Ex: Microsoft and Apple) Find the (totals) 2022, 2021, 2020, and 2019 amounts for the following (ex: Yahoo...

Study smarter with the SolutionInn App


