(a) Using standard reduction potentials from Appendix 11, determine values of f G(K + , aq)...
Question:
(a) Using standard reduction potentials from Appendix 11, determine values of ΔfGº(K+, aq) and ΔfGº(F‾, aq).
(b) Hence, find ΔsolGº(KF, s) at 298 K, if ΔfGº(KF, s) = –537.8 kJ mol–1.
(c) What does the value for ΔsolGº(KF, s) imply about the solubility of KF in water?
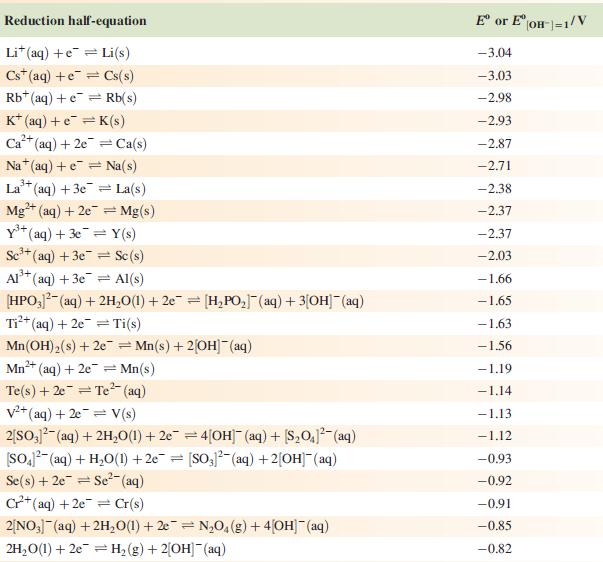
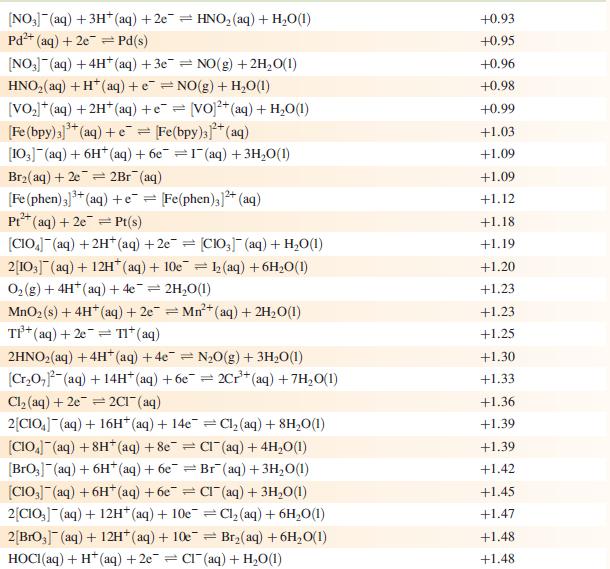
Data from Appendix 11
The concentration of each aqueous solution is 1 mol dm−3 and the pressure of a gaseous component is 1 bar (105 Pa). (Changing the standard pressure to 1 atm (101 300 Pa) makes no difference to the values of Eo at this level of accuracy.) Each half-cell listed contains the specified solution species at a concentration of 1 mol dm−3; where the half-cell contains [OH]−, the value of Eo refers to [OH−] = 1 mol dm−3, hence the notation Eo[OH−] = 1


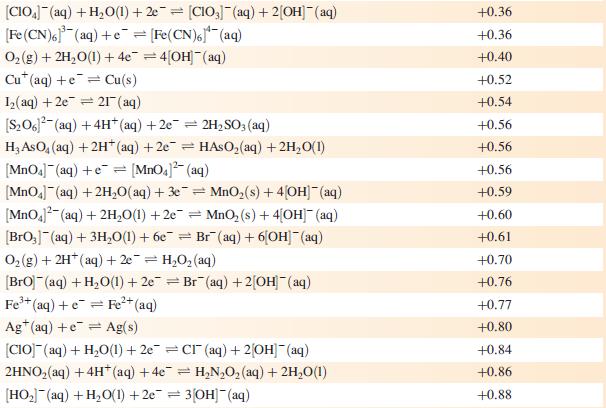

Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





