Assign the lines in the UV photoelectron spectrum of CO shown in Fig. 2.31 and predict the
Question:
Assign the lines in the UV photoelectron spectrum of CO shown in Fig. 2.31 and predict the appearance of the UV photoelectron spectrum of the SO molecule.
Figure 2.31.
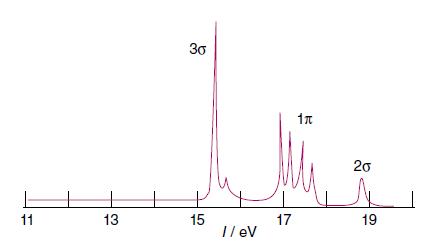
Transcribed Image Text:
11 13 30 15 // eV 17 1π 20 19
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
In Fig 231 the UV photoelectron spectrum of CO is shown The photoelectron spectrum provides informat...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The photoelectron spectrum of CO has four major peaks with ionization energies of 14.5, 17.2, 20.1, and 38.3 eV. Assign these peaks of molecular orbitals of CO, and prepare a quantitative energy...
-
The photoelectron spectrum of CO has four major peaks with ionization energies of 14.5, 17.2, 20.1, and 38.3 eV. Assign these peaks of molecular orbitals of CO, and prepare a quantitative energy...
-
The 1H NMR spectrum of 1-chloropentane is shown at 60 MHz (spectrum H) and 500 MHz (spectrum I), Explain the differences in appearance of the two spectra, and assign the signals to specific hydrogens...
-
Coca-Cola is seeking: To determine the effect of age on intention to purchase Coke Zero; To examine the effect of price on intention to purchase Coke Zero. What are the independent variable(s) and...
-
E-Eyes.com Bank just issued some new preferred stock. The issue will pay an annual dividend of $20 in perpetuity, beginning 20 years from now. If the market requires a return of 5.8 percent on this...
-
Two isolated rock outcrops that share similar characteristicssequence of layers and fossil communitiesare separated by 50 km. Could these two outcrops be related? What principle helps to confirm...
-
Taj Mahabub was the founder and CEO of GenAudio, a Colorado-based audio technology company. GenAudio had struggled financially practically since its formation, and Mahabub wished to secure a...
-
Ashley's Auto Top Carriers currently maintains plants in Atlanta and Tulsa that supply major distribution centers in Los Angeles and New York. Because of an expanding demand, Ashley has decided to...
-
Multiple Choice Question One indication of financial weakness is that Multiple choice question. the fair value of the firm's debt is higher than the book value. current cash from operations is more...
-
What are the expected changes in bond order and bond distance that accompany the following ionization processes? (a) 0 0; +e; (b) N +eN; (c) NO-NO+ + e
-
Assume that the MO diagram of IBr is analogous to that of ICl (Fig. 2.24). (a) What basis set of atomic orbitals would be used to generate the IBr molecular orbitals? (b) Calculate the bond order of...
-
4 / 9x = 5
-
Why would managers not consider drastically modifying their company's strategy?
-
We learned about various ways that leaders/managers experience pressures from stakeholder groups that may have different needs (e.g., HBR, 1991). Discuss how you would respond when various groups or...
-
1. While managing operations there is nothing more important than the safety of any organizations' employees. Workers must be provided a work environment that is safe and free of foreseen hazards. To...
-
A $1,000 par bond with a 4% semi-annual coupon has 15 years to maturity trades at a yield of 6%. What would be its price?
-
What are the multifaceted challenges posed by workplace diversity, encompassing factors such as cultural differences, varying perspectives, and inclusion dynamics, and how do they influence...
-
For each situation below identify the population and the sample and identify p and p if appropriate and what the value of p is. Would you trust a confidence interval for the true proportion based on...
-
The polar coordinates of a point are given. Find the rectangular coordinates of the point. (-1, - /3)
-
ReO 3 is a structure-prototype. Each Re(VI) centre is octahedrally sited with respect to the O 2 centres. The unit cell can be described in terms of a cubic array of Re(VI) centres, with each O 2...
-
Give explanations for the following observations. (a) Raising the temperature of a sample of -Fe from 298K to 1200K (at 1 bar pressure) results in a change of coordination number of each Fe atom from...
-
Comment on the structural and compositional implications of (a) The Fe-deficiency of iron(II) oxide, (b) The anion-excess nature of uranium(IV) oxide.
-
Answer the following questions: 1. Prepare a list of any four (4) methods of extending and developing individual creative thinking skills. 2. Identify and document two (2) tools you can use to extend...
-
1. Explain why all costs are controllable by someone at some point, but in the short run, some costs may be classified as noncontrollable. 2. What does it mean to have a favorable variance? 3. What...
-
Labor Unions answer the following questions: What are the advantages and disadvantages of labor unions? Are unions more popular or less popular now than they have been historically? Why?

Study smarter with the SolutionInn App


