Derive a CBC classification for: (a) W(CO)6 5 (b) (n-Cp)2 WH2 (c) WMe6 (d) (15-Cp)Fe (e) (n-C6H6)Cr
Question:
Derive a CBC classification for:
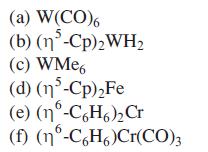
Transcribed Image Text:
(a) W(CO)6 5 (b) (n³-Cp)2 WH2 (c) WMe6 (d) (15-Cp)₂Fe (e) (nᵒ-C6H6)₂Cr (f) (nº-C6H6)Cr(CO)3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To derive the CBC 18electron classification for the given compounds we need to count the total number of valence electrons contributed by each atom in ...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1. Name the FLSA classification (covered or exempt) for each position in the table below, and give your analysis for each classification. 2. Suppose that Falstaff is a plant manager and exercises his...
-
Cruger Bike Company (CBC) makes the frames used to build its bicycles. During 2014, CBC made 20,000 frames; the costs incurred follow. Unit-level materials costs (20,000 units x $30) ....... $...
-
CBC is a government owned enterprise which is legislated to generate its own income which it uses to cover capital and recurrent expenditures. The legislations also give the government the powers to...
-
McIntyre Industries Work in Process Inventory account had a $68,000 beginning balance on May 1 ($40,000 of this related to direct materials used during April, while $28,000 related to conversion...
-
The comparative balance sheets for Yanik Company as of December 31 are presented below. Additional information: 1. Operating expenses include depreciation expense of $42,000. 2. Land was sold for...
-
Repeat Problem 19.16 with the damper partially closed at 10, 20, and 30. Repeat Problem Compute the pressure drop as 1500 cfm of air flows through a wide-open damper installed in a 16-in-diameter...
-
Tom Bonacci brought his Jeep to Brewer Service Station to investigate a strange noise the vehicle was making. The Jeep was raised up on an automobile lift so that Brewer employee Paul Gebing could...
-
Deleon Inc. is preparing its annual budgets for the year ending December 31, 2013. Accounting assistants furnish the data shown below. An accounting assistant has prepared the detailed manufacturing...
-
What are the capabilities of online analytical processing (OLAP)? What is the value of this tool to business? Is OLAP much different from data mining and warehousing? Explain..!
-
(a) The reaction between BiCl 3 and 3 equivalents of EtMgCl yields compound X as the organo product. Two equivalents of BiI 3 react with 1 equivalent of X to produce 3 equivalents of compound Y. In...
-
Draw a bonding scheme (similar to that in Fig. 24.7b) for the interaction of an 3 -allyl ligand with a low oxidation state metal centre. Figure 24.7b. Vacant spda Filled yz M Vacant Filled 8 (b)
-
Share Repurchase in the previous problem, suppose apply Pie has announced it is going to repurchase $6,000 worth of stock. What effects will this transaction have on the equity of the firm? How many...
-
With verifiable fundamens and references, how can the compensation program help a company achieve its strategic goals?
-
Explain the organisational structure of the WTO. Do you agree that the dispute settlement mechanism is more efficient under the WTO than it was under the GATT?
-
Summarize a brief description of human resources management web site, the available resources and your opinion of the benefit of the site to you as a current or future business management/human...
-
The concave mirror with a focal length of 10 cm is placed in front of a pen, with the pen located at the center. The concave mirror is to the left of the pen. A converging lens has a focal length of...
-
.What are the limitations of the rational decision-making model? Give an example. . What is another model of decision-making that offers a better solution for this limitation? Give an example. ....
-
Nancy, the owner of a very successful hotel chain in the Southeast, is exploring the possibility of expanding the chain into a city in the Northeast. She incurs $35,000 of expenses associated with...
-
State whether each of the following will increase or decrease the power of a one-way between-subjects ANOVA. (a) The effect size increases. (b) Mean square error decreases. (c) Mean square between...
-
Assuming that H o f is constant in the interval 275 K600. K, calculate G o for the process (H 2 O, g, 298 K) (H 2 O, g, 600.K). Calculate the relative change in the Gibbs energy.
-
When 2-chloro-1, 1, 2, 3, 3-pentamethylcyclohexane is treated with sodium hydroxide, neither E2 nor S N 2 products are formed. Explain.
-
Why are the triple point temperature and the normal freezing point very close in temperature for most substances?
-
The K a of benzoic acid is 6.30 10 -5 . The pH of a buffer prepared by combining 50.0 mL of potassium benzoate and 50.0 mL of 1.00 M benzoic acid is?
-
Required information [The following information applies to the questions displayed below.] The following post-closing trial balance was drawn from the accounts of Little Grocery Supplier (LGS) as of...
-
Sulfur dioxide is converted to sulfur trioxide over a V2O5 catalyst at a pressure of 1.5 atm. SO2 + O2 SO3 In the reactor configuration in figure below, 100 mol/min feed with composition of 11%...

Study smarter with the SolutionInn App


