Discuss the bonding between the central p-block elements in the following compounds and give the expected arrangements
Question:
Discuss the bonding between the central p-block elements in the following compounds and give the expected arrangements of the organic substituents with respect to the central E2-unit:
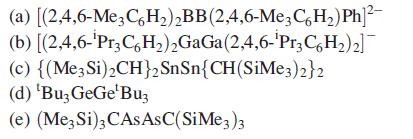
Transcribed Image Text:
(a) [(2,4,6-Me3C6H₂)₂BB (2,4,6-Me3C6H₂) Ph]²- (b) [(2,4,6-'Pr3C6H₂)2GaGa (2,4,6-'Pr3C6H₂)2] (c) {(Me3 Si)₂CH}2 (d) 'Bu3 GeGe¹Buz (e) (Me3 Si)3 CASASC(SiMe3)3 SnSn{CH(SiMe3)2}2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To discuss the bonding between the central pblock elements in the given compounds we need to identify the central elements E and their coordination en...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
The NMR spectra below are for the organic compounds C6H12 and C4H10O. Deduce the structures for these compounds. See Exercise 70 for a discussion of the bonding in organic compounds. The structure of...
-
Two franchising experts recently debated the issue of whether new college graduates should consider franchising as a pathway to entrepreneurship. Jeff Elgin said recent college graduates are not...
-
Suppose Columbia Sportswear Company had accounts receivable of $299,585,000 at January 1, 2014, and $226,548,000 at December 31, 2014. Assume sales revenue was $1,244,023,000 for the year 2014. What...
-
A line source of sodium light ( 0 = 589.3 nm) illuminates a Lloyds mirror 10.0 mm above its surface. A viewing screen is 5.00 m from the source and the whole apparatus is in air. How far apart are...
-
Almetals, Inc., a Michigan company, entered into a contract with the German firm Wickeder Westfalenstahl regarding the purchase of clad metal, a specialty metal used in a variety of industries but...
-
In preparing for the upcoming holiday season, Fresh Toy Company (FTC) designed a new doll called The Dougie that teaches children how to dance. The fixed cost to produce the doll is $100,000. The...
-
Topic : SQL DDL & DML Purpose The purpose of this assignment is to help you learn more about SQL. Brief Introduction SQL is a domain-specific language used in programming and designed for managing...
-
Whether the bonding in lithium alkyls is predominantly ionic or covalent is still a matter for debate. Assuming a covalent model, use a hybrid orbital approach to suggest a bonding scheme for (MeLi)...
-
Write down formulae for the following ions: (a) Manganate(VII); (b) Manganate(VI); (c) Dichromate(VI); (d) Vanadyl; (e) Vanadate (ortho and meta); (f) Hexacyanidoferrate(III). Give an alternative...
-
A pressure-measuring device has been analyzed and can be described by a model with the structure shown in figure a. In other words1 the device responds to pressure changes as if it were a first-order...
-
A balanced die is rolled 180 times. Let be the number of cases when die shows the number 4 on its top face. (a) Find #x (b) Find o (c) Use normal approximation to binomial to approximate P(35 x 40).
-
Ms. Moth dies on February 1, 2024. All of her income is from employment activities and she does not have a spouse or common-law partner. What is the latest date for filing her 2023 income tax return...
-
Let x, J, J, J IR 21 a) Prove that x (J (U U )) = (x J ) - ( 3) J3 U3 Hint: Use cofactor development arguments b) Prove that: (x x J) (J x Js) = det J X X. J J J 1
-
Communities at the margin, minority groups, underserved areas and other groups as integral parts of any city and place of residence should be benefactors of development activities and aspirations...
-
Suppose the following is a possible distribution of the number of crashes a driver will have over the course of their lifetime. X P(X = x) 0 0.01 1 2 (b) Find P(X < ) or P(X> ). P(x < )= P(x > )=...
-
Dan Simms is the president and sole shareholder of Simms Corporation, 1121 Madison Street, Seattle, WA 98121. Dan plans for the corporation to make a charitable contribution to the University of...
-
If the joint cost function for two products is C(x, y) = xy2 + 1 dollars (a) Find the marginal cost (function) with respect to x. (b) Find the marginal cost with respect to y.
-
You get a PT phase diagram by projecting a PVT phase diagram on the PT plane.
-
The mechanism of the following transformation involves a carbocation intermediate that rearranges in a way that we have not yet seen. Rather than occurring via a methyl shift or a hydride shift, a...
-
An ideal gas is expanded adiabatically into a vacuum. Decide which of q, w, U and H is positive, negative, or zero.
-
Use the function, g(x)=|x|. Move the function to the left by 4 units, up by 6 units, stretch vertically by a factor of 3 units, and reflect over the y-axis.
-
The function l(s)=0.046s^(2)-.199s+.264 . If a skid mark with a length of 82.5ft was found, find the speed of the car. Round your answer to the nearest tenth.
-
A light is hung 15 ft above a straight horizontal path. If a man 6 feet tall is walking away from the light at the rate of 5 f(t)/(s), how fast is his shadow lengthening?

Study smarter with the SolutionInn App


