Discuss the trends in stabilities of oxidation states for Group 16 elements at pH = 0, as
Question:
Discuss the trends in stabilities of oxidation states for Group 16 elements at pH = 0, as represented by the Frost diagram in Fig. 16.5, and state the likely nature of the species prevailing for each entry.
Figure 16.5.
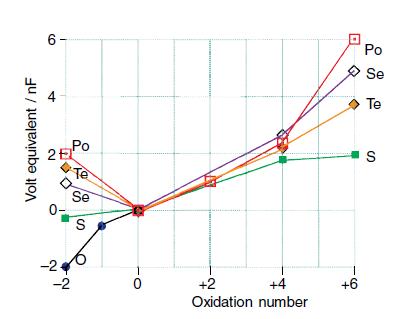
Transcribed Image Text:
Volt equivalent / nF 6- 25 Po E -2 Te Se S 1 0 +2 +4 Oxidation number +6 Po Se Te S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Trends in Stabilities of Oxidation States at pH 0 Oxygen O Oxygen is highly electronegative and has a strong tendency to gain electrons making the hig...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The structures of another class of high-temperature ceramic superconductors are shown below. a. Determine the formula of each of these four superconductors. b. One of the structural features that...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
The Gap Inc. is a global specialty retailer operating stores selling casual apparel, personal care, and other accessories for men, women, and children under The Gap, Banana Republic, and Old Navy...
-
The Bay City Parks and Recreation Department is considering building several new facilities, including a gym, an athletic field, a tennis pavilion, and a pool. It will base its decision on which...
-
Harmony Industries Inc. is a small manufacturer of electronic musical instruments. The plant manager received the following variable factory overhead report for the period: The plant manager is not...
-
We Are Social and Hootsuite reported that the typical American spends 2.02 hours (121 minutes) per day accessing the Internet via mobile devices. In order to test the validity of this statement, you...
-
A European recession and the U.S. economy a. In 2014, European Union spending on U.S. goods accounted for \(18 \%\) of U.S. exports (see Table 17-2), and U.S. exports amounted to \(15 \%\) of U.S....
-
Clearcast Communications Inc. is considering allocating a limited amount of capital investment funds among four proposals. The amount of proposed investment, estimated income from operations, and net...
-
MaxBiotech Inc., a Japanese pharmaceutical manufacturer, has agreed to pay JPY 35.5 billion to Pfizer for the right to use its patent on the coronavirus vaccines. This payment is classified as a...
-
A mechanistic study of reaction between chloramine and sulfite has been reported (B.S. Yiin, D.M. Walker, and D.W. Margerum, Inorg. Chem., 1987, 26, 3435). Summarize the observed rate law and the...
-
In November 2006 the former KGB agent Alexander Litvinenko was found to have been poisoned by radioactive polonium-210. Write a review of the chemical and radiological properties of Po and discuss...
-
When food is irradiated with gamma rays from a cobalt-60 source, does the food become radioactive? Defend your answer.
-
When an object is accelerating, what do you know about its momentum and the forces acting on it? Describe why athletes learn to bend their knees or loosen their joints during high intensity impacts...
-
Correct the errors in the following code so the function returns "yhappyh" without changing the return line of code. def string_change(): = my_string "happy" mult_string=my_string * 2...
-
The Puerto Rico Supervision, Administration and Economic Stability Act (PROMESA) Explains what each of them was (or is). * Why do they arise? * What is the purpose of each of them? * What have been...
-
Taxpayer owned stock in InvestCo. On January 1 she notified InvestCo that all future dividends were to be paid to her son. InvestCo paid $5,000 in dividends during the year. Who must include the...
-
According to guest speaker Melissa Silber, financial ratios, more specifically your debt to equity ratio on your balance sheet, is important to know for the purposes of getting potential funding from...
-
In a random sample of males, it was found that 23 write with their left hands and 217 do not. In a random sample of females, it was found that 65 write with their left hands and 455 do not (based on...
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
In the complex [Ti(BH 4 ) 3 (MeOCH 2 CH 2 OMe)], the Ti(III) centre is 8-coordinate. Suggest modes of coordination for the ligands.
-
Comment on the variation in oxidation states of the first row metals.
-
Write out, in sequence, the first row d-block elements and give the valence electronic configuration of each metal and of its M 2+ ion.
-
Both Personal Selling and Advertising are among the oldest and most effective forms of promotion. Using three ( 3 ) criteria, compare advertising and personal selling as marketing communication...
-
In cell W15: Use the appropriate nested functions: If the employee's Year of Hire is 1974 or 1977 or 1987, and the Region is RG2 or RG4, and the Gender is Male, multiply the Annual Units Sold by the...
-
amounts in millions, except per share data NET SALES Cost of Sales January 31, 2016 Fiscal Year Ended(1) February 1, 2015 February 2, 2014 $ 88,519 $ 83,176 $ 78,812 58.254 54.787 51,897 30,265...

Study smarter with the SolutionInn App


