Each compound in List 1 has a matching description in List 2. Correctly match the partners. There
Question:
Each compound in List 1 has a matching description in List 2. Correctly match the partners. There is only one correct statement for each compound.
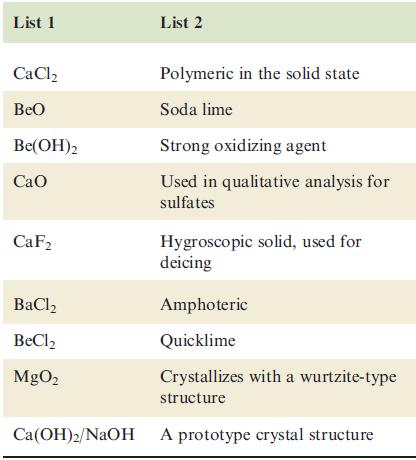
Transcribed Image Text:
List 1 CaCl₂ BeO Be(OH)2 Cao CaF2 List 2 BaCl, BeCl₂ MgO₂ Polymeric in the solid state Soda lime Strong oxidizing agent Used in qualitative analysis for sulfates Hygroscopic solid, used for deicing Amphoteric Quicklime Crystallizes with a wurtzite-type structure Ca(OH)2/NaOH A prototype crystal structure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
It seems you have a list of compounds in List 1 and their corresponding descriptions in List 2 The t...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A small consulting company wants to make the switch to QuickBooks Online. All of their expenses are paid at the time they are incurred, so there is no need to track bills in the system. There is only...
-
Find the equation of the tangent line and the normal line to the curve the x-intercept closest to the origin. f(x)=x-3x+at
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Kamloops Company is a grocery wholesaler and is planning to expand its operations. The company has asked the bank for a loan to finance the expansion. Alphonzo, the companys manager, has prepared the...
-
Mercury Skateboard Company manufactures skateboards. Several weeks ago, the firm received a special-order inquiry from Venus, Inc. Venus desires to market a skateboard similar to one of Mercurys and...
-
During a visit to a plastic sheeting plant, it was observed that a 60-m-long section of a 2-in nominal (6.03-cm-outer-diameter) steam pipe was extended from one end of the plant to the other with no...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the IRR(s) for this project. b. Is this project economically attractive? EOY 0 1 2 3 4 5 6 NCF -$101 $411...
-
Edington Electronics Inc. produces and sells two models of pocket calculators, XQ-103 and XQ-104. The calculators sell for $12 and $25, respectively. Because of the intense competition Edington...
-
You are to PAY 350,000 on March 16. Todays spot rate is $1.15/. The forward rate is $1.18/. On March 16 the spot rate is $1.14/. Futures contracts are for 125,000 each. Q1. How many contracts do you...
-
(a) What qualitative test is used for CO 2 ? (b) What reaction takes place (c) What is observed in a positive test?
-
Suggest products for the following reactions, and write balanced equations for the reactions. Comment on any of these reactions that are important in chemical manufacturing processes. (a) CaH + HO ...
-
The Parallel Axis theorem also holds for lines that are perpendicular to a lamina. Use this fact to find the moment of inertia of the rectangular lamina of Problem 24 about an axis per pendicular to...
-
Liz Claiborne, like other well-known apparel makers, embarked on a major product expansion strategy in the 1990s when it acquired many smaller branded clothing and accessory companies and started...
-
Four consumers are willing to pay these amounts for haircuts: Each firm can give, at most, one haircut. To achieve efficiency, how many haircuts should be given? Which businesses should cut hair and...
-
Colgate-Palmolive Company operates two product segments. Go to the company Web site (http://www.colgatepalmolive.com), and then click on the For Investors link. From there, go to the SEC filings and...
-
Pick an organization, such as a local travel agency or supermarket. Describe its organizational domain, then draw a map of the forces in its general and specific environments that affect the way it...
-
Develop a fault tree for events that could lead to release of benzene to the atmosphere for the distillation section designed in problem 9.5. Data from problem 9.5 A distillation column separates...
-
Fairchild Centre is an NFPO funded by government grants and private donations. It was established on January 1, Year 5, to provide counseling services and a drop in centre for single parents. On...
-
g(x) = x 5 5x 6 a. Show that g(x) = 0 has a root, , between x = 1 and x = 2. b. Show that the equation g(x) = 0 can be written as x = (px + q) 1/r , where p, q and r are integers to be found. The...
-
Name and classify the following hydrogen compounds: (a) BaH 2 , (b) SiH 4 , (c) NH 3 , (d) AsH 3 , (e) PdH 0.9 , (f) HI.
-
Which hydrogen bond would you expect to be stronger, SHO or OHS? Why?
-
Describe the expected physical properties of water in the absence of hydrogen bonding.
-
1A) Out of the four building blocks (efficiency, quality, innovation, customer service)to competitive advantages, on which ones can Starbucks claim strength (better than peers)? 1B) Identify at least...
-
Describe how business models for stakeholders in the health industry are (or are not) built for optimized patient outcome?
-
Should hero pay for essential workers during the COVID-19 pandemic be considered a reward or an incentive? Why?

Study smarter with the SolutionInn App


