For a given atom Y, the YF bond is usually stronger than the corresponding YCl bond. An
Question:
For a given atom Y, the Y—F bond is usually stronger than the corresponding Y—Cl bond. An exception is when Y is oxygen (Table 16.2). Suggest a reason for this observation.
Table 16.2.
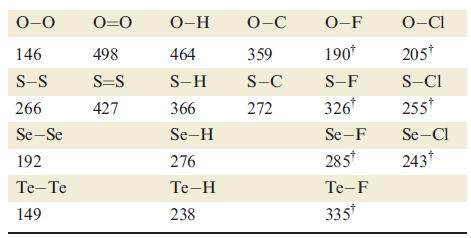
Transcribed Image Text:
0-0 146 S-S 266 Se-Se 192 Te-Te 149 0=0 498 S=S 427 O-H 464 S-H 366 Se-H 276 Te-H 238 O-C 359 S-C 272 O-F 190* S-F 326* Se-F 285* Te-F 335* O-CI 205* S-CI 255* Se-Cl 243†
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
The reason for the observation that the YF bond is stronger than the corresponding YCl bond for most ...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When a beam of atoms emerges from an oven at the absolute temperature T, the most probable de Broglie wavelength for a given atom is In this expression, m is the mass of an atom, and k is Boltzmann's...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Figure shows a cycle consisting of five paths: AB is isothermal at 300 K, BC is adiabatic with work = 5.0 J, CD is at a constant pressure of 5 atm, D E is isothermal, and EA is adiabatic with a...
-
Identify several ways in which you currently use accounting information in your life as a student. Also identify several situations in which, while you are still a student, you might be required to...
-
What is maximum tolerable downtime (MTD)?
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the IRR(s) for this project. b. Is this project economically attractive? EOY 0 1 2 3 4 5 6 NCF -$101 $411...
-
Hamilton County Parks is planning to develop a new park and recreational area on a recently purchased 100-acre tract. Project development activities include clearing playground and picnic areas,...
-
16. Brenda's Balloon Shop is a price taker, and the table below shows its costs of production. Use it to answer the following questions: Output (balloons Total Cost (dollar per hour) per hour) 0 4 1...
-
Briefly discuss the trends in boiling points and values of vap H listed in Table 17.2 for the hydrogen halides. Table 17.2. Property Physical appearance at 298 K Melting point /K Boiling point/K...
-
[NS 2 ][SbF 6 ] reacts with nitriles, RCN, to give [X] [SbF 6 ] where [X] + is a cycloaddition product. Propose a structure for [X]+ and show that it is a 6-electron system. Do you expect the ring to...
-
a. A batch of 50 parts contains six defects. If two parts are drawn randomly one at a time without replacement, what is the probability that both parts are defective? b. If this experiment is...
-
Sunland Corporation began operations on January 1, 2025. The following information is available for Sunland on December 31, 2025. Accounts receivable $1,020 Supplies expense 2,660 Equipment 12,740...
-
Current Attempt in Progress Joseph Taylor recorded the following transactions during the month of April. Apr. 3 16 20 Cash Service Revenue Rent Expense Cash Salaries and Wages Expense Cash 3,020 690...
-
5 ints eBook Print References The following financial statement information is from five separate companies. Beginning of year Assets Liabilities End of year Assets Liabilities Changes during the...
-
8- The Y Company has bought 20.000 worth of commodities but did not pay the money yet. How to record this transaction? A) B) C) D) Commodities 20.000 Cash 20.000 Commodities 20.000 Account's Payable...
-
3. [%6] You are considering two retransmission strategies for all hosts in a network using the ALOHA protocol. The first is a deterministic strategy where the first time a collision is detected, the...
-
Explain how we test H0: = 0 versus Ha: 0. What do we conclude if we reject H0: = 0?
-
How can NAFTA be beneficial to suppliers of Walmart?
-
Aluminosilicate surfaces in zeolites act as strong Brnsted acids, whereas silica gel is a very weak acid. (a) Give an explanation for the enhancement of acidity by the presence of Al 3+ in a silica...
-
(a) Give two examples of applications of quantum wells. (b) Describe why quantum wells are used and if either molecular materials or traditional solid-state materials can exhibit similar properties....
-
Propose formulas for structures that would be isomorphous with SiO 2 and zeolites of the same stoichiometry involving Al, P, B, and Zn, or mixtures thereof, replacing Si.
-
Rico is excited about the revenues that Orion generated during its first year of business. The main revenue source is a matchmaking service for clients seeking a romantic relationship. New clients...
-
3 marks] 9. An open-top box is to be constructed from a rectangular piece of cardboard measuring 52 cm by 36 cm. The box is created by cutting congruent corners and then folding up the sides. (Use...
-
Q1: Which accounting assumption details that a company will continue to exist long enough or for an indefinite period to meet its objectives and commitments and will not liquidate for the foreseeable...

Study smarter with the SolutionInn App


