For each of the following complexes, give the oxidation state of the metal and its d n
Question:
For each of the following complexes, give the oxidation state of the metal and its dn configuration:
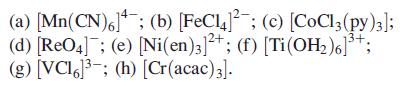
Transcribed Image Text:
(a) [Mn(CN)6]; (b) [FeC¹4]¯¯; (c) [CoCl3 (py)3]; (d) [ReO4]; (e) [Ni(en)3]2+; (f) [Ti (OH₂)6]³+; (g) [VCI6³; (h) [Cr(acac) 3].
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
To determine the oxidation state of the metal and its d electron configuration in each complex we need to apply some general principles of coordinatio...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
You have decided to purchase additional memory for your computer in order to better support the latest version of the Windows operating system. At the local computer store, you notice that not only...
-
Give the number of (valence) d electrons associated with the central metal ion in each of the following complexes (a) K3[Fe(CN)6], (b) [Mn(H2O)6](NO3)2, (c) Na[Ag(CN)2], (d) [Cr(NH3)4Br2]ClO4, (e)...
-
Give the outer electron configuration for each of the following columns in the periodic table. 1) 3A Express your answer as a string without blank space between orbitals. For example, the outer...
-
A taxpayer had the following income: a.) Gain on sale of domestic stocks - P200,000............ b.) Gain on sale of foreign bonds - P100,000............ c.) Gain on sale of a commercial lot in...
-
Order the following major concepts that have helped define the OSCM field on a time line. Use 1 for the earliest to be introduced, and 5 for the most recent. _______Supply chain management...
-
Using Eqs. (7.9), (7.10), and (7.11), show that the resultant of the two waves and is = E01 sin [wt k(x + Ax)] E1 %3D E2 = E01 sin (wt kx)
-
If a well-behaved investment alternative's internal rate of return (IRR) is equal to MARR, which of the following statements about the other measures of worth for this alternative must be true? 1....
-
Nayak Company has recorded the following items in its financial records. Cash in bank ............ $ 41,000 Cash in plant expansion fund ...... 100,000 Cash on hand ............ 8,000 Highly liquid...
-
The figure depicts Jack - in - the - box: "Jack" is attached inside a box by a spring, as shown. You estimate Jack's mass to be 0 . 4 k g . As so often happens, a sign tells you the spring constant:...
-
The vibrational modes of KrF 2 are at 590, 449 and 233 cm 1 . Explain why only the bands at 590 and 233 cm 1 are observed in the IR spectrum of gaseous KrF 2 .
-
By referring to the following literature source, assess the safety precautions required when handling XeO 4 : M. Gerken and G.J. Schrobilgen (2002) Inorg. Chem., vol. 41, p. 198.
-
Based on the financial statements provided for Coca-Colain the Cohesion Case, how much dividends in dollars did Coke pay in 2016? In 2017?
-
If a firm has two divisions and one division is riskier than the other, what would be the potential result if the firm used its overall WACC to evaluate the projects in both divisions? Multiple...
-
Assume that we use a perpetual inventory system and that five identical units are purchased at the following four dates and costs: April 5 $10 April 10 $12 April 15 $14 April 20 $16 April 22 $17 One...
-
Question 10 Marla borrows $4,500 and repays the loan with four equal quarterly installments, with the first installment made 3 months after the loan is made. If interest is charged at a rate of 12...
-
Males and females are observed to react differently to a given set of circumstances. It has been observed that 70% of the females react positively to these circumstances, whereas only 40% of males...
-
Would you consider Glen to be suffering from depression? How did Glen's counsellor assess him for suicide risk?
-
Alison owns a painting that she received as a gift from her aunt 10 years ago. The aunt created the painting. Alison has displayed the painting in her home and has never attempted to sell it....
-
(a) What do data breach notification laws require? (b) Why has this caused companies to think more about security?
-
As explained previously, the concentration of an alcohol can be selected such that both a broad signal and a narrow signal appear simultaneously. In such cases, the broad signal is always to the...
-
For each of the following IR spectra, identify whether it is consistent with the structure of an alcohol, a carboxylic acid, or neither. a. b. c. d. e. f. 100- 80- 60- 40 20- 0. 2000 4000 3500 3000...
-
For each of the following IR spectra, determine whether it is consistent with the structure of a ketone, an alcohol, a carboxylic acid, a primary amine, or a secondary amine. a. b. c. d. e. f. 100-...
-
Write a script that will do the following [1 -1 1 1- A=1 1 -1 [1 -1 1 2- Find the rank of A 3- Find Determinant of A 4- Find Borth(A). In Matlab, the command orth(A) provides a set of orthonormal...
-
What is the output? int x = 8; while (x > 0) { System.out.printf("%d",x); x = x/2; }
-
4. What is the binary value being output from Y in the multiplexer circuit shown to the right? 0 D D Dy Y S So

Study smarter with the SolutionInn App


