From the relative atomic orbital and molecular orbital energies depicted in Fig. 3.32, describe the character as
Question:
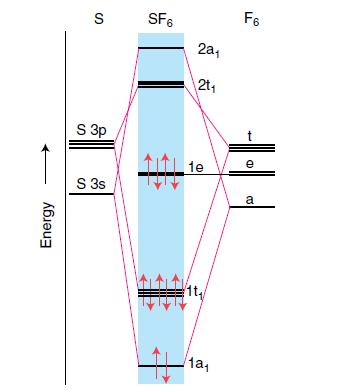
From the relative atomic orbital and molecular orbital energies depicted in Fig. 3.32, describe the character as mainly F or mainly S for the frontier orbitals e (the HOMO) and 2t (the LUMO) in SF6. Explain your reasoning.
Figure 3.32.
Transcribed Image Text:
Energy S SF6 S 3p S 3s H 221 2t₁ 1e 1a₁ F6 e a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To answer this question we need to interpret the relative atomic orbital and molecular orbital energ...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The FEMO theory (Problem 11.14) of conjugated molecules is rather crude and better results are obtained with simple Huckel theory. (a) For a linear conjugated polyene with each of N carbon atoms...
-
Molecular orbitals are most commonly delocalized throughout the molecule and exhibit distinct bonding or anti-bonding character. Loss of an electron from a specific molecular orbital from excitation...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Accounting for Contingencies and Provisions In accounting, contingencies and provisions play a crucial role in preparing financial statements and ensuring accurate representation of a company's...
-
You own a portfolio that has $2,950 invested in Stock A and $3,700 invested in Stock B. If the expected returns on these stocks are 8 percent and 11 percent, respectively, what is the expected return...
-
A wave pulse moves along a strip of rubber with a velocity of 10 cm/s as depicted in Figure P12.66. Five equally spaced points are marked on the rubber strip labeled A though E as shown.? (a) Rank...
-
On March 13, 1963, Ernesto Miranda was arrested for kidnapping and rape and taken to a Phoenix police station. After being identified by the complaining witness, he was questioned by two police...
-
Stratton Testing is considering investing in a new testing device. It has two options: Option A would have an initial lower cost but would require a significant expenditure for rebuilding after 5...
-
Problem 4. Suppose that A in an m x n matrix and b is a vector in R. Let a = (a1,..., am) be a vector in Rm. Carefully prove that the following two statements are equivalent: There is a vector x R"...
-
(a) Determine the point group of the PF 5 molecule. (Use VSEPR, if necessary, to assign geometry.) (b) What is the maximum degeneracy of its molecular orbitals? (c) Which P3p orbitals contribute to a...
-
Consider the p orbitals on the four Cl atoms of tetrahedral [CoCl 4 ] 2 , with one p orbital on each Cl pointing directly at the central metal atom. (a) Confirm that the four p orbitals which point...
-
Cameron is a newborn baby whose parents are thinking about vaccinating him against disease. The figure illustrates the marginal benefits and marginal costs of vaccinations for Cameron. Cameron's...
-
The following information was derived from the 2017 accounting records of Swifty Corporation: Swifty's Central Warehouse Swifty's Goods Held by Consignees Beginning inventory $128000 $ 14000...
-
Beta of Assets A, B, C, and D are 1.2, 1.8, 2.4, and 0.8 respectively. You form a portfolio with equal weights on A, B, C, and D. The risk-free rate is 2%, and the expected return of the market...
-
Jewel is also looking to expand this project to its store on the South side. If the expected cash flows are as follows, what calculate the crossover rate (where the manager is indifferent between...
-
What are the options available to deal with Toyota's chip shortage problems and challenges ? Explain
-
3. Given the following table detailing labor and capital use in a particular country, Exports Imports Capital ($ million) $3.55 $5 Labor (person-years) 192 160 Capital/Labor ($/person) a. Under...
-
Comment on the value of the formula (EFR) approach to estimating funding requirements. Could it create more problems than it solves?
-
Write an SQL statement to display all data on products having a QuantityOnHand greater than 0.
-
Calculate the corresponding wavelengths of electromagnetic radiation with frequencies of (a) 3:0 10 12 Hz, (b) 1:0 10 18 Hz (c) 5:0 10 14 Hz. By referring to Appendix 4, assign each wavelength or...
-
Hydrogen possesses three isotopes, but tritium ( 3 H), which is radioactive, occurs as less than 1 in 10 17 atoms in a sample of natural hydrogen. If the value of A r for hydrogen is 1.008, estimate...
-
Arsenic is monotopic. What does this statement mean? Using Appendix 5, write down three other elements that are monotopic. Data from Appendix 5 Element Actinium Aluminium Americium Antimony Argon...
-
Who is responsible when AI goes wrong? The robot or the company? Provide a business example of a time that AI has "gone wrong". Discuss the impact this mishap had on the company and/or its customers.
-
Given all the information you have been provided by Samantha and the city planner, and Samantha's desired revenue, would you recommend that Zeplins open the new location, and why would you make this...
-
Think of a problem encountered in the past by a group of which you are a member. Begin your analysis by defining the group. What type of group was it? At what stage of development was the group? And...

Study smarter with the SolutionInn App


