How do you account for the fact that, although potassium is placed after argon in the periodic
Question:
How do you account for the fact that, although potassium is placed after argon in the periodic table, it has a lower relative atomic mass?
Transcribed Image Text:
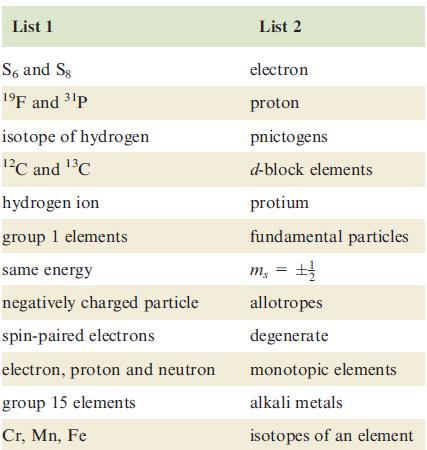
List 1 S6 and Sg 1⁹F and ³¹p isotope of hydrogen ¹²℃ and ¹³℃ hydrogen ion group 1 elements same energy negatively charged particle spin-paired electrons electron, proton and neutron group 15 elements Cr, Mn, Fe List 2 electron proton pnictogens d-block elements protium fundamental particles m₁ = ±1/ allotropes degenerate monotopic elements alkali metals isotopes of an element
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
The apparent discrepancy between the placement of potassium and argon in the periodic table and thei...View the full answer

Answered By

Kratika Agrawal
I have already worked as an subject experts.
Hello! After reviewing your assignment criteria, I am certain than I can provide the greatest for you by the deadline. This assignment is the area of my specialization kindly considers my proposal and get a quality paper. Thanks!
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
IST 8100 Supplemental Cases Inside Microsoft During last fall's United Way campaign at Microsoft, two vice-presidents made a wager on whose division would generate the most contributions. The loser,...
-
In what ways does the clinical approach to selection differ from the statistical approach? How do you account for the fact that one approach is superior to the other?
-
A uniform marble rolls down a symmetric bowl, starting from rest at the top of the left side. The top of each side is a distance h above the bottom of the bowl. The left half of the bowl is rough...
-
5 Melbourne Corporation has traditionally made a subcomponent of its major product. Annual production of 30,000 subcomponents results in the following costs: Direct materials Direct labor Variable...
-
Accounting has its own vocabulary and basic relationships. Match the accounting terms at the left with the corresponding phrase at the right. ____ 1. Posting ____ 2. Normal balance ____ 3. Payable...
-
A family spends $46,000 a year for living expenses. If prices increase by 3 percent a year for the next three years, what amount will the family need for their living expenses after three years?
-
Maribel Baltazar was hired by clothing retail merchandiser Forever 21 in 2007. During the hiring process, Baltazar was given an 11-page document to sign, two pages of which contained an arbitration...
-
Lori, age 28, is a registered nurse who earns $4000 monthly working in a hospital. She is seriously injured in an auto accident in which she is at fault and is expected to be unable to work for at...
-
Write a MATLAB program that asks the user to enter his results in four quizzes (q1,q2, q3, and q4) out of 100, and calculate the average value. Then print the output given in the Table below to the...
-
For a neutral atom, X, arrange the following atomic orbitals in an approximate order of their relative energies (not all orbitals are listed): 2s, 3s, 6s, 4p, 3p, 3d, 6p, 1s.
-
Write down the six sets of quantum numbers that describe the electrons in a degenerate set of 5p atomic orbitals. Which pairs of sets of quantum numbers refer to spin-paired electrons?
-
The methylation of DNA, the acetylation of histones, and the packaging of DNA into chromatin by certain kinds of proteins are sometimes referred to as epigenetic modifications of the DNA. These...
-
How do changes in planned expenditures affect the aggregate demand curve?
-
What can increase the equilibrium interest rate in the liquidity preference framework?
-
The definition of the government deficit is a matter of debate. What would be the effect on the measurement of the government deficit if one considered Social Security taxes a forced loan to the...
-
What condition is required for equilibrium in the money market? Why does the money market move toward equilibrium?
-
What are open market operations? How does the Fed use these operations to increase or decrease the money supply?
-
Exhibit 9-1 is an extract from the University of Saskatchewans 2012/13 annual report. (Most post-secondary institutions in Canada have a fiscal year that ends on March 31, so it includes transactions...
-
The manager for retail customers, Katie White, wants to hear your opinion regarding one business offer she has received from an entrepreneur who is starting a mobile phone app called Easy Money. The...
-
Explain why compounds of beryllium are mainly covalent whereas those of the other Group 2 elements are predominantly ionic.
-
Marble and limestone buildings are eroded by contact with acid rain. Define the term acid rain and discuss the origins of the acidity. Describe the processes by which the marble and limestone are...
-
Explain how the nature of the alkyl group affects the structure of lithium alkyls.
-
Find the equation of the line between the points (2, 3) and (4,-1)
-
Find f(g(x)) and g(f(x)) and determine whether the pair of functions f and g are inverses of each other. f(x)=x-2 and g(x)=x+2 f(g(x)) =
-
Express-6-121 as a complex number (in terms of i): -6-121 =

Study smarter with the SolutionInn App


