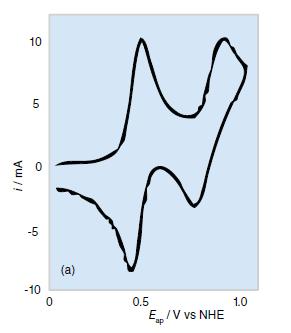
How would the cyclic voltammetry shown in Fig. 8.53a differ if (a) The Os(IV) complex decomposed rapidly;
Question:
How would the cyclic voltammetry shown in Fig. 8.53a differ if
(a) The Os(IV) complex decomposed rapidly;
(b) Os(III) is oxidized in a single, rapid two-electron step to Os(V)?
Figure 8.53a.
Transcribed Image Text:
i/mA 10 LO 5 O -5 -10 0 (a) 0.5 Ep/V vs NHE ap 1.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a If the OsIV complex decomposed rapidly the second wave in the cyclic voltam...View the full answer

Answered By

RASEENA CK
computer science degree completed at calicut university an done yeardegree of library science at ignou university.I am worked in private education centre by tutoring students miainly my subject is mathematics.and i conducted other subject like computr physics
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
To carefully prepared mitochondria were added succinate, oxidized cytochrome c, ADP, orthophosphate, and sodium cyanide. Referring to Figure 14.16, answer the following. (a) List the sequence of...
-
Is an isothermal process necessarily internally reversible? Explain your answer with an example.
-
Presented below is information related to Alexis and Ryans, Attorneys at Law. Retained earnings, January 1, 2015 ......$ 23,000 Legal service revenue2015 ......... 340,000 Total expenses2015...
-
A financial analyst uses the following model to estimate a firms stock return: Return = 0 + 1 PE + 2 P/S + , where P/E is a firms price-to-earnings ratio and P/S is a firms price-to sales ratio....
-
In spring 1989, Michael Jordan and the Chicago Bulls were in Indianapolis, Indiana, to play against the Indiana Pacers. At the same time, Karla Knafel was singing with a band at a hotel in...
-
Cost Behavior and Cost-Volume-Profit Analysis for Many Glacier Hotel The purpose of this integrated exercise is to demonstrate the interrelationship between cost estimation techniques and subsequent...
-
the tuition fee for maths tuition for john is expected to be 6500 per year if he attend tuition for 4 year starting from 4 years from now how much does he have to have in her account which yields 6...
-
Sketch the forms of the following solution phase NMR spectra (abundances shown as per cent): (a) The 1 H-NMR spectrum of KBH 4 (b) The 1 H- and 195 Pt-NMR spectra of cis-[Pt(CO) 2 (H)Cl] 1 H, I = ,...
-
Determine the g values of the EPR spectrum shown in Fig. 8.57, measured for a frozen sample using a microwave frequency of 9.43 GHz. Figure 8.57 300 340 380 420 B/mT 460 500
-
This question deals with the properties of possible worlds, defined on page 488 as assignments to all random variables. We will work with propositions that correspond to exactly one possible world...
-
Direct financing allows investors to easily: a. Diversify their investment portfolio b. Lower search and transaction costs c. Expand their portfolios d. Match amounts and maturity of...
-
Here is a Java programming question: Note: Use s.hasNextLine for multiple lines and terminate the input with "end" Don't use string.split or arrays, just use flow controls, loops and iterations. The...
-
What is negative carry? What does this mean and why is it important to a hedging strategy?
-
Write a formal business letter, to be sent out to one of your stakeholders congratulating them on completing 10 years of business together. the business letter must have a practical application, so...
-
Mortgage-backed securities are believed to have contributed to the recent subprime mortgage crisis. Assess the fairness of this statement, providing a rationale for your reasoning and a...
-
Calculate the wavelength of light emitted when each of the following transitions occur in the hydrogen atom. What type of electromagnetic radiation is emitted in each transition? a. n = 4 n = 3 b. n...
-
What is an insurable interest? Why is it important?
-
How would you attempt to (a) Estimate the crystal field stabilization energy of FeF 2 , (b) Determine the overall stability constant of [Co(NH 3 ) 6 ] 3+ in aqueous solution given that the overall...
-
Suggest the formula and structure of the mononuclear complex formed between Cr 3+ and ligand 21.83. Comment on possible isomerism. CO N CO N (21.83) CO
-
(a) Which of the following complexes would you expect to suffer from a JahnTeller distortion: [CrI 6 ] 4 , [Cr(CN) 6 ] 4 , [CoF 6 ] 3 and [Mn(ox) 3 ] 3 ? Give reasons for your answers. (b) [Et 4 N] 2...
-
1) Jenna gets a ticket for careless driving after the police come to investigate a car accident she had with you on Hanover Boulevard. Your car is badly damaged through no fault of your own. Is Jenna...
-
What is the 10 plus 2 rule . How does it affect the companys global transportation operations?
-
Write a program to calculate the total price for car wash services. A base car wash is $10. The cost for additional services are as follows: Air freshener Rain repellent Tire shine Wax Vacuum $1 $2...

Study smarter with the SolutionInn App


