Identify the metal-containing compounds A, B, C, and D in this scheme: Ba- HO - A HO
Question:
Identify the metal-containing compounds A, B, C, and D in this scheme:
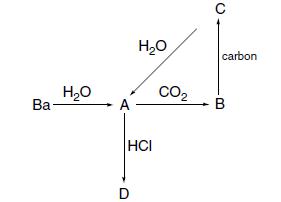
Transcribed Image Text:
Ba- H₂O - A H₂O НСІ D CO₂ carbon B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To identify the metalcontaining compounds A B C and D in the given scheme lets analyze the prov...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
A, B, C and D are all adult males and skilled musicians. (A plays guitar, B bass, C keyboards and D drums.) All have worked previously as professional musicians. In January 2019, A invites B, C and D...
-
Identify each of the compounds A-D in the reaction scheme shown in Fig. P26.67, p. 1331. Explain your answers. H,N NH NaNO /HCI CH(CH3)2 HCL H20 C -heat (CHs)-CH-CH-=O + D
-
Restaurants have traditionally used bottom-end wines to sell by the glass (BTG) at reasonably low prices per glass. In recent years, there has been a growing trend within the restaurant industry to...
-
If the resultant force acting on the hook is F R = {? 200i + 800j + 150k} lb, determine the magnitude and coordinate direction angles of F. 30 F= 600 ib
-
Bach Company makes household water filtration equipment. The Vinson Division manufactures filters. The Vine Water Division then uses the filters as a component of the final product Bach sells to...
-
How could an investor use XBRL to do an analysis of the industrys inventory turnover?
-
Mr. Tam is the front man for the Asian American dance-rock band The Slants. Mr. Tam named his band The Slants to reclaim and take ownership of Asian stereotypes. The band draws inspiration for its...
-
Answer the following multiple-choice questions: a. In 2007 and 2008, Zoret Company reported earnings per share of $0.80 and $1.00, respectively. In 2009, Zoret Company declared a 4-for-1 stock split....
-
The current in the 1 3 . 8 2 resistor is / 3 = 0 . 9 A . 1 . Find the current in the other resistors in thecircuit? 2 . What is the total energy dissipated in resistor 4 1 7 . 2 0 if the power supply...
-
The molecular geometries of crown ether derivatives play an important role in capturing and transporting alkali metal ions. K. Okano and co-workers (see K. Okano, H. Tsukube, and K. Hori,...
-
Account for the fact that LiF and CsI have low solubility in water whereas LiI and CsF are very soluble.
-
Find each of these values? a) (133 mod 23 + 261 mod 23) mod 23 b) (457 mod 23 182 mod 23) mod 23
-
What do the terms risk bearer, provider, payer, and practitioner relationship signify within the context of healthcare and insurance industries, elucidating their respective roles and interactions?
-
Your position is that of an Operations Analyst. You will be asked to make several calculations and solve problems for this product line. Your skills will be gained through the material in this...
-
How to implement a training plans and communication plans in health disparity in rural communities?
-
ABC purchased 21% stock, the cost value of 1,000,000, the parent company held this investment in non controlling activity. How could ABC benefit if non controlling interest was reported as a...
-
Boston Corporation received $70,000 cash from issuance of notes payable. How is this to be reported on the statement of cash flows?
-
When the supply of oxygen is limited, iron metal reacts with oxygen to produce a mixture of FeO and Fe 2 O 3 . In a certain experiment, 20.00 g of iron metal was reacted with 11.20 g of oxygen gas....
-
Show that the peak of the black body spectrum as a function of ? is given by eq. (22.14) kg T Wmax = 2.82
-
Determine the shapes of each of the following molecules and then, using the data in Table 2.2, state whether each is expected to be polar or not: (a) H 2 S; (b) CO 2 ; (c) SO 2 ; (d) BF 3 ; (e) PF 5...
-
Draw Lewis structures for (a) CO 2 , (b) SO 2 , (c) OF 2 (d) H 2 CO.
-
Draw the resonance structures for the O 3 molecule. What can you conclude about the net bonding picture?
-
Bob and Larry have been working at their organization for the same amount of time, and they seem to work equally hard at their work and contribute about the same amount to their organization. Bob has...
-
Can someone please help me with the below..they need to be journal entries. Any help is very much appreciated! You realize that the $3,000 paid by Teacher's College on 1/27 (INV-1011) was for a...
-
you should choose any topic and write the outline by following this way: Introduction Thesis: State in a full sentence your topic, what categories you used for your criteria to judge it, and your...

Study smarter with the SolutionInn App


