In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. (a) Predict the
Question:
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed.
(a) Predict the structure of SF4 using the VSEPR model.
(b) Account for the fact that at 298K and in solution the 19F NMR spectrum of SF4 exhibits a singlet but that at 175 K, two equal-intensity triplets are observed.
Table 4.3
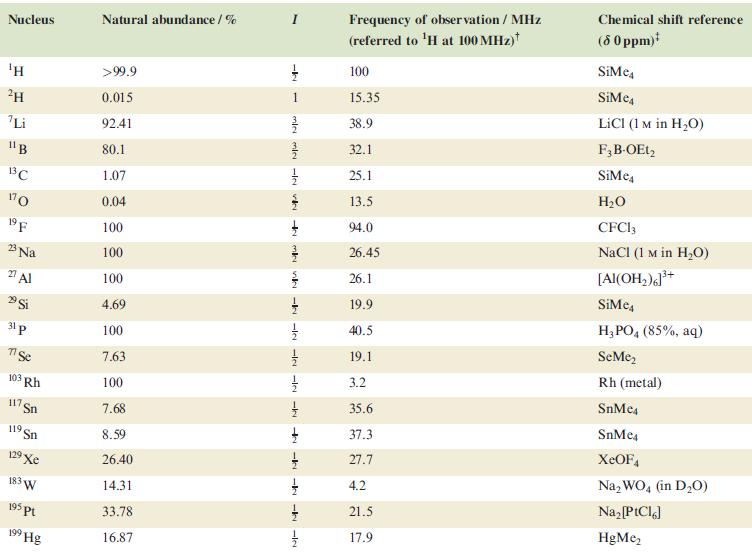
Transcribed Image Text:
Nucleus ΤΗ ²H Li 11B 13C 170 19 F 23 Na 2 Al 29 Si 31 p 7 Se 103 Rh 117. Sn 119 Sn 19 Xe 183 W 195 pt 199 Hg Natural abundance/% >99.9 0.015 92.41 80.1 1.07 0.04 100 100 100 4.69 100 7.63 100 7.68 8.59 26.40 14.31 33.78 16.87 miele v + mk nh -k -le -le-le - -ki -k -k -IN Frequency of observation / MHz (referred to ¹H at 100 MHz) 100 15.35 38.9 32.1 25.1 13.5 94.0 26.45 26.1 19.9 40.5 19.1 3.2 35.6 37.3 27.7 4.2 21.5 17.9 Chemical shift reference (8 0 ppm)* SiMe SiMe4 LICI (1 M in H₂O) F3B-OEt2 SiMe H₂O CFC13 NaCl (1 M in H₂O) [Al(OH₂)]³+ SiMc4 H3PO4 (85%, aq) SeMe₂ Rh (metal) SnMc4 SnMe4 XeOF4 Na₂WO4 (in D₂O) Na₂ [PtCl6] HgMe₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a To predict the structure of SF using the VSEPR model Valence Shell Electron Pair Repulsion model we first determine the valence electron count of the sulfur S and fluorine F atoms Sulfur is in group ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
VSEPR (valence state electron pair repulsion) theory was formulated to anticipate the local geometry about an atom in a molecule (see discussion in Section 25.1). All that is required is the number...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. NaBH 4 contains the tetrahedral [BH4] ion. Although NaBH 4 hydrolyses slowly in water, it is possible to obtain a...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Rationalize the fact that the 13 C NMR spectrum of CF 3 CO 2 H consists of two binomial quartets with coupling...
-
Two forces are acting on the 5 kg object, with only one of them shown in Fig. 1. Knowing that F = 20 N, 0 = 30 and that the object is moving at a constant acceleration of a = 12 m/s, find the second...
-
Jake needed a summer job and was lucky enough to land a position as a ticket collector at a local amusement park. On his first day, he was assigned to work alongside Tim who had worked at the park...
-
Itemized deductions are taken when a. The taxpayer wants to. b. They are less than the standard deduction. c. They are higher than the standard deduction. d. The standard deduction is limited by high...
-
Crush Autosmashers can purchase a new electromagnet for moving cars at a cost of \($20,000.\) At the end of its useful life, the electromagnet will be worth \($1,000.\) If Crushs MARR is 12...
-
Jake owns a lawn maintenance company, and Luke owns a machine repair shop. For the month of July, the following transactions occurred. July 3 Jake provides lawn services to Lukes repair shop on...
-
Amazon, Inc. Presentation Your chief executive officer (CEO) has asked you to present the company's (Amazon, Inc.) process on making decisions under risks and uncertainty at the annual shareholders'...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The structure of [P5Br 2 ] + is shown in diagram 4.17. Account for the fact that the 31 P NMR spectrum of this...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Outline the mechanism of Berry pseudo-rotation, giving two examples of molecules that undergo this process. Table...
-
Give the IUPAC names of the following alcohols. (a) (b) (c) (d) (e) (f) ,, OH Br CH3 , H,C Cl - CH,CH2 CH,OH CH,CH,CH H-OH CH3
-
Summarize the position that a dividend policy may be irrelevant with regard to the firms stock price.
-
What is the basic controversy surrounding capital structure theory?
-
What is the typical frequency with which cash dividends are paid to investors?
-
Distinguish among the (a) declaration date, (b) date of record, (c) ex-dividend date.
-
Explain the different types of value.
-
Jijang Excavations Ltd. (JEL) operates specialized equipment for installing natural gas pipelines. JEL, which has a December 31 year end, began 2016 with a single piece of equipment that had been...
-
On 1 July 2018, Parent Ltd acquired all the shares of Son Ltd, on a cum-div. basis, for $2,057,000. At this date, the equity of Son Ltd consisted of: $ 1,000,000 Share capital 500 000 shares...
-
When equal volumes of nitric oxide (NO) and air are mixed at atmospheric pressure a rapid reaction occurs, to form NO 2 and N 2 O 4 . However, nitric oxide from an automobile exhaust, which is...
-
Are reactions of NO 2 as an oxidizing agent generally faster or slower when pH is lowered? Give a mechanistic explanation for the pH dependence of NO 2 oxidations.
-
Write the balanced chemical equation corresponding to the standard enthalpy of formation of P 4 O 10 (s). Specify the structure, physical state (s, l, or g), and allotrope of the reactants. Does...
-
How much less is a perpetuity of $2,000 worth than an annuity due of the same amount for 30 payments (in $ dollars)? Assume an interest rate of 10%. $
-
A bond has a face value of $4000 and matures on June 21 2025 with a bond rate of 6.2% compounded semi-annually. If the bond was purchased on December 21 2019 at a market rate of 6.9% compounded...
-
Suppose Wesley Publishing's stock has a volatility of 55%, while Addison Printing's stock has a volatility of 20%. If the correlation between these stocks is 75%, what is the volatility of the...

Study smarter with the SolutionInn App


