In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The 19 F
Question:
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed.
The 19F NMR spectrum of each of the following molecules exhibits one signal. For which species is this observation consistent with a static molecular structure as predicted by the VSEPR model:
(a) SiF4;
(b) PF5;
(c) SF6;
(d) SOF2;
(e) CF4
Table 4.3
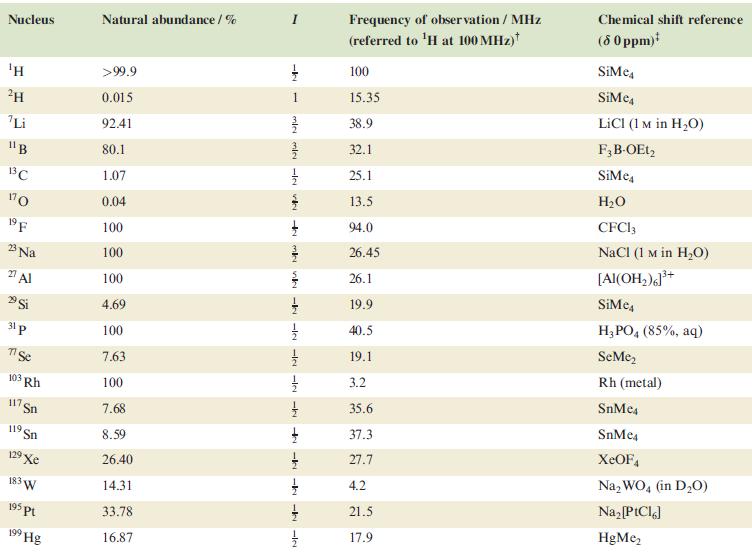
Transcribed Image Text:
Nucleus ΤΗ ²H Li 11B 13C 170 19 F 23 Na 2 Al 29 Si 31 p 7 Se 103 Rh 117. Sn 119 Sn 19 Xe 183 W 195 pt 199 Hg Natural abundance/% >99.9 0.015 92.41 80.1 1.07 0.04 100 100 100 4.69 100 7.63 100 7.68 8.59 26.40 14.31 33.78 16.87 miele v + mk nh -k -le -le-le - -ki -k -k -IN Frequency of observation / MHz (referred to ¹H at 100 MHz) 100 15.35 38.9 32.1 25.1 13.5 94.0 26.45 26.1 19.9 40.5 19.1 3.2 35.6 37.3 27.7 4.2 21.5 17.9 Chemical shift reference (8 0 ppm)* SiMe SiMe4 LICI (1 M in H₂O) F3B-OEt2 SiMe H₂O CFC13 NaCl (1 M in H₂O) [Al(OH₂)]³+ SiMc4 H3PO4 (85%, aq) SeMe₂ Rh (metal) SnMc4 SnMe4 XeOF4 Na₂WO4 (in D₂O) Na₂ [PtCl6] HgMe₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
To determine which species have a static molecular structure as predicted by the VSEPR model we need to consider the number of unique fluorine environ...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
19F is the only isotope of fluorine that occurs naturally, and it has a nuclear spin of + (a) Into how many peaks will the proton signal in the 1H NMR spectrum of methyl fluoride be split? (b) Into...
-
19F is the only isotope of fluorine that occurs naturally, and it has a nuclear spin of 1/2 (a) Into how many peaks will the proton signal in the 1H NMR spectrum of methyl fluoride be split? (b) Into...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. (a) In the 1 H NMR spectrum of compound 4.21, there is a triplet at 3.60 ppm (J 10.4 Hz). Assign the signal and...
-
Cadmium is a highly toxic substance with lethal effects on the human body in concentrations greater than 4.4 x 10-mol/L. Cadmium (II) carbonate has a Kp of 5.2 x 10-12 at 25C. Calculate the...
-
Describe the major differences between US GAAP and International Financial Reporting Standards.
-
Of the cereal grass species, only maize contains two copies of each block of linked genes. What does this duplication of sets of maize genes indicate about the origin of this agronomically important...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 334A? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
A large retailer obtains merchandise under the credit terms of 1/15, net 45, but routinely takes 60 days to pay its bills. Given that the retailer is an important customer, suppliers allow the firm...
-
In December of 2017, the US Government signed the Tax Cuts and Jobs Act (TCJA) into law. The TCJA had four goals; tax relief for middle-income families, simplification for individuals, economic...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Outline the mechanism of Berry pseudo-rotation, giving two examples of molecules that undergo this process. Table...
-
1.0mmol of the ligand, L, shown below was reacted with 0.50 mmol of PtCl 2 . The positive mode MALDI-TOF mass spectrum of the purified product was run in -cyano-4-hydroxycinnamic acid matrix. The...
-
What is an ad-hoc query?
-
How is a Eurobond different from a bond issued in Asia that is denominated in dollars?
-
Distinguish between the residual dividend theory and the clientele effect.
-
A construction company called Alians Group is considering buying new machinery for one of its projects. Leasing Company, which deals in heavy machinery, owns that particular equipment and is looking...
-
What managerial logic might lie behind a stock split or a stock dividend?
-
What does the current yield tell us?
-
Canada Canning Company owns processing equipment that had an initial cost of $106,000, expected useful life of eight years, and expected residual value of $10,000. Depreciation calculations are done...
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
Explain why compounds of Bi(III) typically have the metal atom in a distorted geometry.
-
Identify the compounds A, B, C, and D. As Cl D A C LIAIH4 Cl/hv 3 RMgBr B
-
Use the VSEPR model to predict the probable shapes of (a) PCl 4 + , (b) PCl 4 , (c) AsCl 5 , (d) SbF 5 2 , (e) SbF 6 .
-
(Present value of an uneven stream of payments) You are given three investment alternatives to analyze. The cash flows from these three investments are as follows: End of Year 1 2 3 4 5 Investment A...
-
A 10-year, 9% coupon bond currently sells for $92. Another 10-year currently sells for $84 and pays half the coupon rate as the first bond. What is the 10-year zero rate? (Please enter the rate in...
-
Eric is planning to save $47.17 every month for 4 years. He plans to make his first savings contribution in 1 month from today. If he currently has $901.00 saved and expects to have $3,981.10 in 4...

Study smarter with the SolutionInn App


