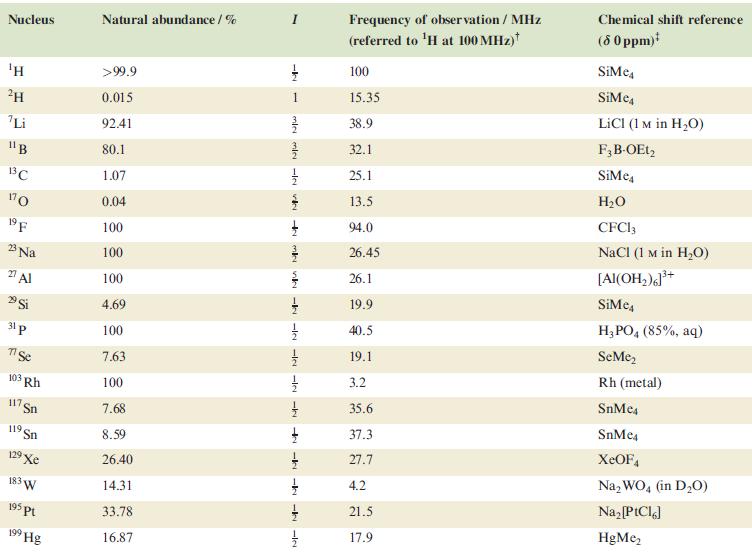
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Figure 4.39 shows
Question:
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed.
Figure 4.39 shows the 11B NMR spectra of
(a) THF·BH3 (4.16)
(b) PhMe2P·BH3.
Interpret the observed coupling patterns and mark on the figure where you would measure relevant coupling constants.
Figure 4.39
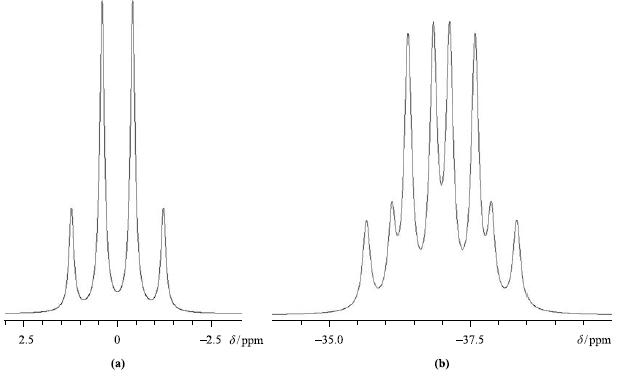
Table 4.3
Transcribed Image Text:
0-BH3 (4.16)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
To interpret the observed coupling patterns in 11B NMR spectra we need to analyze the boron environment in the given chemical structures Boron11 11B i...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. (a) Predict what you would expect to see in the 15 N NMR spectrum of the isotopically labelled compound cis-[Pt( 15...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The 19 F NMR spectrum of the octahedral ion [PF5Me] shows two signals ( 45.8 and 57.6 ppm). Why are two signals...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. MeCN solutions of NbCl 5 and HF contain a mixture of octahedral [NbF 6 ] , [NbF 5 Cl] , [NbF 4 Cl 2 ] , [NbF 3 Cl...
-
What are the upper and lower 2.5th percentiles for a chi-square distribution with 2 df? What notation is used to denote these percentiles? Pulmonary Disease The data in Table 6.10 concern the mean...
-
Identify the missing internal control in the following situations. Select from these activities: Proper authorization Separation of duties Adequate documents and records a. While reviewing the...
-
Which of the following items does not qualify as an educator expense item deduction? a. Books. b. Home schooling expenses. c. Computers and software. d. Professional development expenses related to...
-
Using only the factor formulas given in Table 2.6, derive Equation 7.5 starting with Equation 7.3. TABLE 2.6 Summary of Discrete Compounding Interest Factors. To Find Given Factor Symbol Name P F...
-
Installment-Sales Method and Cost-Recovery Method on January 1, 2010, Wetzel Company sold property for $250,000. The note will be collected as follows: $120,000 in 2010, $90,000 in 2011, and $40,000...
-
Recent events have once again shaken and stirred the memory of the nation. Almost immediately after the Civil War, a myth arose painting the conflict with a brush that attempted to blur the lines...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The 29 Si NMR spectrum of compound 4.15 shows a triplet with a coupling constant of 194 Hz. (a) Rationalize these...
-
Indicate the amount (if any) that Michael can deduct as ordinary and necessary business deductions in each of the following situations and explain your solution. a. From time to time, Michael rents a...
-
The current assets and current liabilities sections of the statement of financial position of Agincourt Corp. are as follows: The following errors have been discovered in the corporations accounting:...
-
Assuming the market is efficient, what is the relationship between a stocks price and the security market line?
-
What three actions can a firm take to minimize its net working capital?
-
Is trade credit a permanent, temporary, or spontaneous source of financing? Explain.
-
When we speak of benefits from investing in an asset, what do we mean?
-
George Andronikashvili has a trade credit with balance of $6,000. George signed a special note, which enables him to pay 4 percent of the debt balance each month, which is $240. How many months...
-
Healthlabs Ltd. purchased lab equipment for $18,000 on January 1, 2016. At that time, management determined that the equipment would have a useful life of three years with no residual value and that...
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
Show with an equation why aqueous solutions of NH 4 NO 3 are acidic.
-
A compound containing five-coordinate nitrogen has been characterized (A. Frohmann, J. Riede, and H. Schmidbaur, Nature, 1990, 345, 140). Describe (a) The synthesis, (b) The structure of the...
-
Ammonia can be prepared by (a) The hydrolysis of Li 3 N (b) The high-temperature, high-pressure reduction of N 2 by H 2 . Give balanced chemical equations for each method starting with N 2 , Li, and...
-
You are looking at two 1 0 - year cash flow projections ( in millions, including resale ) for the same property. The top row shows the estimates the seller of the property is presenting. The bottom...
-
Let A = 50 T(u) = T(v) = a Tofu = [ 1 ] and [8] -4 v = and define T: R2 R by T(x) = Ax. Find the images under T of u = 05
-
Torch Industries can issue perpetual preferred stock at a price of $68.00 a share. The stock would pay a constant annual dividend of $5.50 a share. What is the company's cost of preferred stock, rp?...

Study smarter with the SolutionInn App


