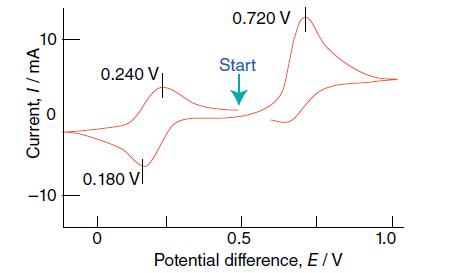
Interpret the cyclic voltammogram shown in Fig. 8.58, which has been recorded for an Fe(III) complex in
Question:
Interpret the cyclic voltammogram shown in Fig. 8.58, which has been recorded for an Fe(III) complex in aqueous solution.
Figure 8.58.
Transcribed Image Text:
10 Current, // mA Currer -10 0.240 V₁ 0.180 VI 0 0.720 V Start 0.5 Potential difference, E / V 1.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The cyclic voltammogram shown in Fig 858 represents the electrochemical behavior of an FeIII complex in an aqueous solution as a function of potential ...View the full answer

Answered By

User l_998468
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Your portfolio consists of $58210 invested in Stock X and $49908 invested in Stock Y. Stock X has a beta of 0.83 and Stock Y has a beta of 1.05. If the return on Treasury bills is 2.0% and the return...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
In Parts I and II of this case, you performed preliminary analytical procedures and assessed acceptable audit risk and inherent risk for Pinnacle Manufacturing. Your team has been assigned the...
-
A railroad flatcar is traveling to the right at a speed of 13.0 m/s relative to an observer standing on the ground. Someone is riding a motor scooter on the flatcar (Fig. 3.43). What is the velocity...
-
Fredonia Repair Inc. was started on May 1. A summary of May transactions is presented below. 1. Stockholders invested $10,000 cash in the business in exchange for common stock. 2. Purchased equipment...
-
Using the information from P6-6A and P6-7A for Veryclear Glassware (California Employer Account Number 999-9999-9), complete the following State of California Form DE-9, Quarterly Contribution and...
-
Fantastic Sams and Defendants PSTEVO, LLC and Jeremy Baker entered into a franchise agreement pursuant to which Fantastic Sams granted PSTEVO a franchise to operate a Fantastic Sams Salon. According...
-
For each of the unrelated transactions described below, present the entry(ies) required to record each transaction. 1. Coyle Corp. issued $10,000,000 par value 10% convertible bonds at 99. If the...
-
A combined solar and auxiliary energy system is used to meet the same load as in Example 12.5. The total cost of the system to cover 65% of the load (solar fraction) is $20,000. The owner will pay a...
-
Which technique is sensitive to the fastest processes, NMR or EPR?
-
Sketch the forms of the following solution phase NMR spectra (abundances shown as per cent): (a) The 1 H-NMR spectrum of KBH 4 (b) The 1 H- and 195 Pt-NMR spectra of cis-[Pt(CO) 2 (H)Cl] 1 H, I = ,...
-
If a circular register buffer is used to handle local variables for nested procedures, describe two approaches for handling global variables.
-
A polyamide A-3 flat belt is used to transmit 15 hp under light shock conditions where K = 1.25, and a factor of safety equal to or greater than 1.1 is appropriate. The pulley rotational axes are...
-
This expenditure (formerly general government consumption) includes all current government expenditures for purchases of goods and services (including an employee). a. Capital expenditure b....
-
Steam enters a turbine at 6000 kPa and 600C and exits as a saturated vapor at 0.02 MPa, producing power at a rate of 2000 kW. Changes in kinetic and potential energy of the flowing fluid can be...
-
Assume you are looking to invest $10,000 and you have two friends each of which is offering to sell you a 25% stake in his/her business in exchange for your $10,000. 1) List at least 5 questions you...
-
In an electron microscope, a beam of high energy electrons can knock out core electrons from atoms in the sample. The subsequent relaxation of electrons from higher energy shells to the empty orbital...
-
Consider the following waves representing electromagnetic radiation: Which wave has the longer wavelength? Calculate the wavelength. Which wave has the higher frequency and larger photon energy?...
-
How can a promoter avoid personal liability for pre-incorporation contracts?
-
Comment on each of the following observations. (a) Li 2 TiO 3 forms a continuous range of solid solutions with MgO. (b) When TiCl 3 is heated with concentrated aqueous NaOH, H 2 is evolved.
-
In the complex [Ti(BH 4 ) 3 (MeOCH 2 CH 2 OMe)], the Ti(III) centre is 8-coordinate. Suggest modes of coordination for the ligands.
-
Comment on the variation in oxidation states of the first row metals.
-
+ 2. Specify which operations run best when the list is sorted. Then briefly describe an algorithm to implement each operation using a list organized in the optimal way. (10%) Task a) Find the...
-
for c++ 6. Table 1 is the summary of examination marks for three students. The students are labelled as student[0], student[1] and student[2]. The subjects are labelled as subject[0], subject[1],...
-
Employer Summary: Kelly is the senior finance manager for Achieve Financial Services and has been tasked with hiring a new financial analyst for their growing international portfolio. Many...

Study smarter with the SolutionInn App


