Refer to Table 21.13 and calculate the rate constants for electron transfer in the oxidation of by
Question:
Refer to Table 21.13 and calculate the rate constants for electron transfer in the oxidation of
![]()
by the oxidants
(a)
![]()
(b)
![]()
Comment on the relative sizes of the rate constants.
Table 21.13.
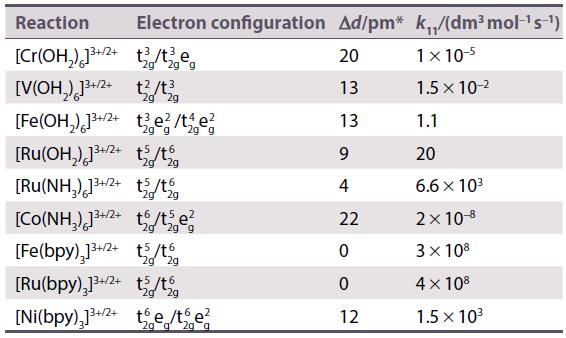
Transcribed Image Text:
[V(OH₂) 1²+ (E° (V³+/V²+) = -0.255V)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Based on the information provided in Table 2113 we can calculate the rate constants for electron tra...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Hansel and Gretel are twins. Gretel goes to a star which is 65 light years away with a constant velocity plane (= v/c) = 0,88. Every birthday, the two do not forget to send each other radio signals,...
-
The photosynthetic reaction centre of the purple photosynthetic bacterium Rhodopseudomonas viridis contains a number of bound cofactors that participate in electron transfer reactions. The following...
-
The equilibrium Ac=' B + C at 25C is subjected to a temperature jump that slightly increases the concentrations of Band C. The measured relaxation time is 3.0 us. The equilibrium constant for the...
-
Define price and name the various types of prices described in this chapter.
-
Ryder Company's balance sheet shows Inventory $162,800. What additional disclosures should be made?
-
A steady-flow adiabatic turbine (expander) accepts gas at conditions T1, P1, and discharges at conditions T2, P2. Assuming ideal gases, determine (per mole of gas) W, Wideal, Wlost, and So for one of...
-
Given a normal random variable , how can we approximate it by a discrete distribution with only two realizations and ? To begin with, given the symmetry of the normal distribution, a natural choice...
-
Match the following terms and descriptions. (1) _____ management assertion (2) _____appropriate (3) _____design and operating (4) _____negligence (5) _____material (6) _____entity level (7)...
-
Suppose over the next period a stock will either go up by a factor u = 1.26, or down by a factor d = 0.76. How many shares of the stock are in the replicating portfolio for the one-period...
-
Solutions of [PtH 2 (PMe 3 ) 2 ] exist as a mixture of cis and trans isomers. Addition of excess PMe 3 led to formation of [PtH 2 (PMe 3 ) 3 ] at a concentration that could be detected using NMR....
-
Write the rate law for formation of [MnX(OH 2 ) 5 ] + from the aqua ion and X . How would you determine if the reaction is d or a?
-
You have an investment that will pay you 1.72 percent per month. How much will you have per dollar invested in one year? In two years?
-
This email is poorly written. Read the weaknesses I have listed and what is needed for improvement. Weaknesses: Fails to provide a meaningful subject line. Does not open directly with a statement...
-
Bribery of government officials commonly is used to obtain business contracts in many foreign countries. This practice is often concealed in company books as "_______." Question 27 options: obtaining...
-
The federal budget has to be approved by Congress. Select one: True False
-
The type of life insurance that pays a death benefit if the policyholder dies within a specific time period, but has no remaining value at the end of the time is called ______________. Group of...
-
Financial Transaction Taxes are wide spread in Zambia Group of answer choices True False
-
In October 1994, a flaw was discovered in the Pentium microchip installed in personal computers. The chip produced an incorrect result when dividing two numbers. Intel, the manufacturer of the...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Give an example of two systems separated by a wall that are in thermal but not chemical equilibrium.
-
A 1.50 mole sample of an ideal gas at 28.5C expands isothermally from an initial volume of 22.5 dm 3 to a final volume of 75.5 dm 3 . Calculate w for this process a. For expansion against a constant...
-
Derive the equation (H/T) V = C V + V/ from basic equations and definitions.
-
Demonstrates the good knowledge of health problem including most of the social determinants and behavioral determinants? Explain briefly
-
FIND THE LEASE RENTAL AND TAX AMOUNT for the four year Year 0 Year 1 Year 2 Year 3 Year 4 Cost of owning Net Purchase price $ 15,00,000 Maintenance Expenses $ 75,000 $ 75,000 $ 75,000 $ 75,000 Tax...
-
How many positive integers between 300 and 599, inclusive 1. are divisible by 3? 2. are divisible by 7? 3. are divisible by 3 and by 7? 4. are divisible by 7 but not by 3? Question 2 We want to split...

Study smarter with the SolutionInn App


