Select the compound on each line with the named characteristic and state the reason for your choice.
Question:
Select the compound on each line with the named characteristic and state the reason for your choice.
(a) Strongest Lewis acid:
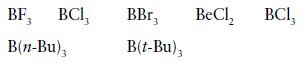
(b) More basic towards B(CH3)3
Transcribed Image Text:
BF, 3 BCI, B(n-Bu), BBr3 B(t-Bu) 3 BeCl₂ BC1₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
To determine the strongest Lewis acid from the given compounds we need to understand what makes a species a strong Lewis acid A Lewis acid is a specie...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
For matrices A and B, find A 2B. A || 32 3 -1 4 0-2 2 B = 145 -1 -2 3
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
For each of the following situations, identify the inventory method that you would use or, given the use of a particular method, state the strategy that you would follow to accomplish your goal: a....
-
Marks 1. Find the limits, if they exist. If a limit does not exist, check whether the function approaches +00 x2 + 2x - 15 (5) (a) lim x-3 x2-4x +3 x2 - 4 (5) (b) lim x2 x4 - 16 Carol Ferland CF...
-
Strauss Company established a predetermined variable overhead cost rate at $10.50 per direct labor hour. The actual variable overhead cost rate was $9.60 per hour. The planned level of labor activity...
-
Determine Ms. As filing status in each of the following independent cases: a. Ms. A and Mr. Z have been living together since 2014. They were married on December 13, 2016. b. Ms. A married Mr. P in...
-
Plaintiff visited South Chicago on January 10, 2008, seeking a new 2008 Nissan Versa (Versa) with manual transmission, anti-lock brakes, and other features. He was told by the employees of South...
-
Tony and Susan are starting a retail business selling formal wear for men and women. They estimate profits and losses for the next five years to be: ($20,000), ($10,000), ($5,000), $10,000, and...
-
Does the monopoly power wielded by professional sports leagues hurt the fans, players, citizens, local economies, or the sport? What solutions could be used ?
-
Draw the structures of chloric acid and chlorous acid and predict their pK a values using Paulings rules.
-
The reversible reaction of CO 2 gas with aqueous emulsions of long-chain alkyl amidine compounds has important practical applications. Describe the chemistry that is involved in this demonstration of...
-
Why is the change in net working capital included in operating cash flow estimates?
-
You are working as a Bookkeeper in your training organisation. The Operations Manager of your training organisation has asked you to prepare a set of financial statements for the current financial...
-
If a customer requests a statement of a brokerage firm's financial condition, what document(s) must the brokerage firm send to the customer immediately?
-
Provide and example of a company's financial decision that could affect the three components of the accounting equation (Assets = Liabilities + Equity)? How might a decision impact assets,...
-
A company buys a $48 machine with a useful life of 3 years. For book purposes, the asset is depreciated using straight-line method. For tax purposes, it's depreciated using MACRS (Yr 1=50%, Yr 2=33%,...
-
A shoe store orders shoes from the manufacturer and sells them at a mall. Storing shoes at the store costs $7 per shoe pair for a year. When reordering shoes from the manufacturer, there is a fixed...
-
Chloral hydrate (C 2 H 3 Cl 3 O 2 ) is a drug formerly used as a sedative and hypnotic. It is the compound used to make Mickey Finns in detective stories. a. Calculate the molar mass of chloral...
-
Transform the while loop from the previous exercise into an equivalent for loop (make sure it produces the same output).
-
The values of pK a (1) and pK a (2) for chromic acid (H 2 CrO 4 ) are 0.74 and 6.49 respectively. (a) Determine values of K a for each dissociation step. (b) Write equations to represent the...
-
(a) Write equations to show how you expect compounds 7.24 to 7.28 to dissociate in aqueous solution. (b) Suggest how compound 7.29 will react with NaOH in aqueous solution. What salts would it be...
-
(a) Two-electron reduction of B 5 H 9 followed by protonation is a convenient route to B 5 H 11 . What structural change (and why) do you expect the B 5 cage to undergo during this reaction? (b)...
-
The essay must be focused on a policy of your choice aimed at mitigating/helping to reduce/stopping some form of criminal activity. The policy may be one that is expected to affect crime indirectly....
-
What three things do businesses typically do when faced with increased risk from cross-border investments?
-
'If women are to have better financial well-being in later life, and reduce potential financially related gender inequalities, then they themselves need to have strong financial capability in early...

Study smarter with the SolutionInn App


