Suggest a structure for the complex [CuCl(19.25)] + assuming that all donor atoms are coordinated to the
Question:
Suggest a structure for the complex [CuCl(19.25)]+ assuming that all donor atoms are coordinated to the Cu(II) centre.
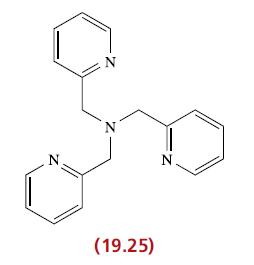
Transcribed Image Text:
N N N (19.25) N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The complex CuCl1925 has one chloride ion coordinated to the copperII center In this case the copper ...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A solution made by dissolving 0.875 g of Co(NH3)4Cl3 in 25.0 g of water freezes at 20.56C. Calculate the number of moles of ions produced when 1 mole of Co(NH3)4Cl3 is dissolved in water and suggest...
-
(a) The electronic absorption spectrum of [Ni(DMSO) 6 ] 2+ (DMSO=Me 2 SO) exhibits three absorptions at 7728, 12970 and 24 038 cm 1 . Assign these absorptions. (b) CuF 2 has a distorted rutile...
-
Suggest a structure for the product of nucleophilic substitution obtained on Solvolysis of tert-butyl bromide in methanol, and outline a reasonable mechanism for its formation.
-
Another equation that has been used to model population growth is the Gompertz equation: dy dt ky In(K/y) , where k and K are positive constants. For each y in 0 < y K, show that dy/ dt is never less...
-
What is meant by a triple-bottom-line strategy? Give an example of a company that has adopted this type of strategy.
-
Consider a molecular motor that consumes the energy from 100 ATP molecules per second. What is the power output of this motor?
-
A snow-cone machine at an icecream shop costs \(\$ 15,000\). The machine is expected to generate profits of \(\$ 2,500\) each year of its 10-year useful life. At the end of the 10 years, the machine...
-
Krauss Leasing Company signs a lease agreement on January 1, 2011, to lease electronic equipment to Stewart Company. The term of the non-cancelable lease is 2 years, and payments are required at the...
-
DATABASE SYSTEMS What is a key and how is it important in a relational model?
-
By referring to relevant sections earlier in the book, write a brief account of the formation of hydrides, borides, carbides and nitrides of the d-block metals.
-
(a) Write down, in order, the metals that make up the first row of the d-block and give the ground state valence electronic configuration of each element. (b) Which triads of metals make up groups 4,...
-
Four years ago, Litho Printers Ltd. purchased a large, four-colour printing press for $450,000 with the intent of using it for 10 years. Recently, the production manager learned that replacing the...
-
1-Your companys revenues were $2 million this year. You paid out $500,000 in salaries and your only other cash outflow was the purchase of a piece of construction equipment for $1 million that is to...
-
Zackie Company has recently allowed its customers to make purchases on credit. The terms are 2/10, n/30. Unfortunately, Zackie Company has found that not all of its credit customers pay their bills...
-
A bullet of mass 20 grams is fired into a pendulum bob of mass 600 grams initially hanging at rest on a string of length 2.0 m. The bullet lodges in the pendulum bob. What minimum speed must the...
-
You have $29,000 in a brokerage account, and you plan to deposit an additional $1,000 at the end of every future year until your account totals $100,000. You expect to earn 10% annually on the...
-
Bristol plc pays corporation tax at a rate of 25%. Tax is paid the year after each cash flow. Please complete the following Calculate the tax cashflows Calculate the net cash flow for each year...
-
Delphinium Company owns two parcels of land ( 1231 assets). One parcel can be sold at a loss of $60,000, and the other parcel can be sold at a gain of $70,000. The company has no nonrecaptured 1231...
-
Suppose the concentration of glucose inside a cell is 0.1 mm and the cell is suspended in a glucose solution of 0.01 mm. a. What would be the free energy change involved in transporting 10-o mole of...
-
For each of the following reactions determine whether ÎS for the reaction (ÎS sys ) will be positive, negative, or approximately zero: a. b. c. d. e. Br Br HCI CI
-
Draw an energy diagram of a reaction with the following characteristics: (a) A one-step reaction with a negative G (b) A one-step reaction with a positive G (c) A two-step reaction with an overall...
-
Predict the major product(s) of the following reaction: Br2 H2S
-
What is the average number of products sold in the last 12 weeks? What was the trend of total and category A and B sales in the previous quarter? (Plot the graph) What can be concluded from the trend...
-
Prepare Wisconsin return for Greg and Mandy James. Their Social Security Numbers are: Greg 412-34-9874 Mandy 587-99-8745 Their address is 8688 Basketball Drive, New Berlin, WI 53151 They have 2...
-
Make the respective entries to the Journal of the different transactions. In each transaction there are the accounts that are affected. The amount of $300.00 of the prepaid rent was used. The amount...

Study smarter with the SolutionInn App


